
-
 Rosenior says Chelsea owners supportive despite slump
Rosenior says Chelsea owners supportive despite slump
-
Oil jumps on Hormuz tensions, stocks retreat

-
 Romania legend Hagi eyes 'winning every game' on return as coach
Romania legend Hagi eyes 'winning every game' on return as coach
-
Rana stars as Bangladesh down New Zealand to level ODI series at 1-1

-
 Real Madrid coach Arbeloa launches stout defence of Mbappe
Real Madrid coach Arbeloa launches stout defence of Mbappe
-
Pope Leo blasts 'exploitation' on visit to resource-rich Angola

-
 Amy Winehouse's father loses suit against friends selling her clothes
Amy Winehouse's father loses suit against friends selling her clothes
-
Japan issues warning after 7.7-magnitude quake hits north

-
 UniCredit woos Commerzbank shareholders in takeover battle
UniCredit woos Commerzbank shareholders in takeover battle
-
European stocks slide as oil jumps on Hormuz tensions

-
 Amy Winehouse's dad loses suit against friends for selling clothes
Amy Winehouse's dad loses suit against friends for selling clothes
-
Slovenian liberal Golob fails to form government

-
 Elon Musk summoned over French X deepfake probe but presence unclear
Elon Musk summoned over French X deepfake probe but presence unclear
-
Tsunami warning as major quake hits northern Japan, shakes Tokyo

-
 Rana takes 5-32 as Bangladesh bowl out New Zealand for 198
Rana takes 5-32 as Bangladesh bowl out New Zealand for 198
-
Anthropic says will put AI risks 'on the table' with Mythos model

-
 Iran says no plan for US peace talks
Iran says no plan for US peace talks
-
Iran executes two more members of exiled opposition: group

-
 Pope Leo visits Angola's diamond-rich northeast
Pope Leo visits Angola's diamond-rich northeast
-
US begins 'biggest ever' Philippines war games in thick of Mideast conflict

-
 Bulgaria ex-president wins parliamentary majority
Bulgaria ex-president wins parliamentary majority
-
US begins 'biggest ever' Philippines war games in thick of Mideast war

-
 Anxiety lingers in divided Kashmir a year after shooting attack
Anxiety lingers in divided Kashmir a year after shooting attack
-
Hit reality show helps rev up Japan's delinquent youth subculture

-
 Oil prices bounce back on Iran war escalation
Oil prices bounce back on Iran war escalation
-
Residents return to ravaged homes months after Hong Kong fire

-
 Australia's Green wins playoff for third LPGA LA Championship title
Australia's Green wins playoff for third LPGA LA Championship title
-
Pakistan's military chief takes lead on US-Iran talks in diplomatic blitz

-
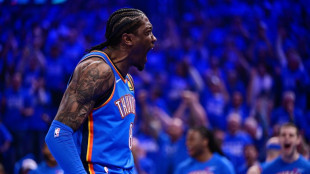 Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
-
US begins Philippines war games in thick of Middle East conflict

-
 Who's Bad? Not Michael Jackson in new big-budget biopic
Who's Bad? Not Michael Jackson in new big-budget biopic
-
Nations gather for first-ever conference on fossil fuel exit

-
 Money, lobbyists, inertia: why fossil fuels are so hard to quit
Money, lobbyists, inertia: why fossil fuels are so hard to quit
-
France summons Elon Musk over X probe

-
 'Save humanity': Four figures battling it out to lead embattled UN
'Save humanity': Four figures battling it out to lead embattled UN
-
Gilgeous-Alexander, Wemby, Jokic finalists for NBA MVP

-
 Israel vows to level homes in Lebanon, counter threats with 'full force'
Israel vows to level homes in Lebanon, counter threats with 'full force'
-
Research Pages Built Around the Metrics Self-Directed Investors Actually Use

-
 BioLargo Engineering, Science & Technologies Awarded $1.2 Million Contract to Design Pilot-Scale Minerals Processing Facility for Resource Recovery
BioLargo Engineering, Science & Technologies Awarded $1.2 Million Contract to Design Pilot-Scale Minerals Processing Facility for Resource Recovery
-
Fast Finance Pay Corp. Launches OK.pay, Advancing OK.secure Into Scalable Payments Ecosystem

-
 MMJ International Holdings Joins Federal Lawsuit on 4/20 to Stop CMS From Introducing Unvalidated Cannabinoid Products Into America's 65+ Population
MMJ International Holdings Joins Federal Lawsuit on 4/20 to Stop CMS From Introducing Unvalidated Cannabinoid Products Into America's 65+ Population
-
Snapshot Recipes AI Mobile App Celebrates Approved Feature-Rich Update and AI Marketing Expansion

-
 GA-ASI Completes MQ-9B 'Flight Into Known Icing' Flight Tests
GA-ASI Completes MQ-9B 'Flight Into Known Icing' Flight Tests
-
Snaplii Reimagines Its Identity and Its Role in Redefining the Future of Finance

-
 Peoples Bancorp Announces First Quarter 2026 Results
Peoples Bancorp Announces First Quarter 2026 Results
-
Elektros Inc. Positioned at the Center of a Multi-Billion Dollar EV Infrastructure Opportunity with Patented Charging Technology

-
 Ondas Receives $10 Million Initial Order, Part of a $50 Million Award, to Launch Large-Scale Border Demining Program Along Israel's Eastern Border
Ondas Receives $10 Million Initial Order, Part of a $50 Million Award, to Launch Large-Scale Border Demining Program Along Israel's Eastern Border
-
Aspire Biopharma Announces Closing of Second and Final Tranche of $21 Million Private Placement by Select Investors; Secures Commitment Letter for $22.5M Credit Facility to Fund the DCS Acquisition

-
 TripSitter Announces Discovery Notice
TripSitter Announces Discovery Notice
-
Global Industrial to Report First Quarter 2026 Results on May 5, 2026


LIXTE's Bold Blueprint to Rewire Cancer's Master Switch and Redefine Its Own Future (NASDAQ: LIXT)
BOCA RATON, FL / ACCESS Newswire / September 16, 2025 / For a small-cap biotech, the headlines often center on fundraising, partnerships, or meeting exchange requirements. But every so often, the spotlight shifts to the science itself. Especially when it's the kind with the potential to rewrite how cancer is treated. That is where LIXTE Biotechnology Holdings (NASDAQ:LIXT) finds itself today.
Earlier this month, the company put the focus squarely on its lead compound, LB-100. It is the only clinical-stage inhibitor of protein phosphatase 2A (PP2A), a cellular enzyme that cancers hijack to survive treatment. That exclusivity is more than a scientific curiosity; it is the kind of value driver that can turn a small-cap biotech into a category creator.
So why does PP2A matter? Think of it as the circuit breaker in cancer's wiring system. When therapies cause DNA damage or stress inside a tumor cell, PP2A shuts things down, giving the cell a chance to recover. Most treatments try to kill cancer directly. LB-100 takes a different approach: it disables the breaker, stripping cancer of its emergency escape route and leaving cells defenseless against therapy.
Cancer's Most Lethal Enemy
Said more directly, LB-100 drives tumors into a death spiral, pushing them into lethal overdrive until their own survival systems collapse. In other words, it becomes cancer's most lethal enemy. By inhibiting PP2A, LB-100 disrupts cancer's ability to repair itself, floods cells with stress signals, and even generates neoantigens that expose tumors to the immune system. In effect, it turns cold, invisible cancers into hot targets for immunotherapy.
This dual punch-forcing self-destruction while making tumors more visible-sets LB-100 apart. It is not just a weapon on its own but an amplifier for existing treatments. When combined with chemotherapy, radiation, or immunotherapy, LB-100 is showing potential to strip away resistance and reveal tumors that were once untouchable.
That is a particularly big deal-better yet, potentially life-saving-for patients with microsatellite stable colorectal cancer, which accounts for about 85% of colorectal cases. For these patients, immunotherapies that transformed other cancer types often fall short. The same is true for ovarian clear-cell carcinoma, another notoriously resistant tumor. LIXTE is betting that LB-100 can change that outcome.
And LIXTE is not making that bet alone.
Global Pharma Partners Join the Mission
Through a partnership with GSK, LIXTE is studying LB-100 in ovarian clear-cell carcinoma alongside Dostarlimab, with GSK funding the trial. At the same time, Roche is providing atezolizumab and trial funding for a study in microsatellite stable colorectal cancer at the Netherlands Cancer Institute. Both programs are designed to answer the essential question: Can LB-100 unlock the effectiveness of leading immunotherapies where they have historically failed?
There are risks, of course. The colorectal study is under a temporary voluntary safety pause following early adverse events, a reminder that novel mechanisms often carry unknowns. Yet Phase 1 data in soft-tissue sarcoma demonstrated LB-100's tolerability with no toxicity concerns. And LIXTE has since discontinued its Phase 2 monotherapy sarcoma program, not as a retreat but as a recalibration. The company is prioritizing immunotherapy combinations where commercial potential is exponentially higher. It is the kind of strategic pruning that signals focus rather than weakness.
In oncology, timing matters. LIXTE knows that drug developers win not by stretching themselves thin but by aiming resources where the science is strongest and the market most receptive. That is why LB-100's journey looks less like a straight line and more like a chessboard, with each move setting up the next. Still, the full weight of LIXTE's story is not in the science alone but in how the company prepares to sustain it.
Where Science Meets Financial Strategy
This summer, LIXTE caught attention for a move that had little to do with clinical trials. It announced the purchase of a $2.6 million portfolio of digital assets, positioning nearly half of its treasury in this new category. For many observers, it was unexpected. For LIXTE, it was intentional.
The company described it as a forward-looking balance sheet strategy designed to create value drivers alongside the science. In an industry where most companies focus exclusively on burning cash, LIXTE is experimenting with how to grow and diversify it. The message was clear: innovation at LIXTE is not confined to the lab. Its treasury can also be an active creator of capital.
Pair that with a leadership team reshaped over the past quarter, including a new CFO and new board members with financial and operational expertise, and you start to see a company aligning its corporate strategy with its scientific ambition. A company developing a first-in-class drug is also building a first-in-class approach to how it funds and sustains that drug.
And that dual-track innovation may prove to be LIXTE's most defining trait.
A Company With Two Narratives
On one level, LIXTE presents the biotech story everyone wants to see: a unique mechanism validated in Nature, clinical collaborations with global leaders like GSK and Roche, and a shot at redefining standards of care in cancers long resistant to treatment. On another level, it is a corporate story about a company unwilling to accept the limits of traditional playbooks, whether in finance, governance, or growth strategy.
In September 2025, those two narratives define LIXTE. Scientifically, it is advancing a one-of-a-kind cancer drug with global validation and partnerships. Strategically, it is reshaping its balance sheet to think differently about value creation. Taken together, LIXTE is showing that real progress comes not from a single dimension but from strength across science, partnerships, and capital.
In oncology, where the margin between success and failure can define lives, that breadth of vision may prove to be the company's defining value driver.
Sources and references:
Company Profile
LIXTE Biotechnology Holdings, Inc. is a clinical-stage pharmaceutical company developing a new class of cancer therapy called PP2A inhibitors. The Company's innovative approach enhances the efficacy of both chemotherapy and immunotherapy, potentially providing new treatment options for patients. At the core of the Company's therapy is LB-100, the Company's proprietary compound that acts as an inhibitor of the PP2A phosphatase with a favorable toxicity profile. LB-100 promotes the production of neoantigens and cytokines, boosts T-cell proliferation, and disrupts the DNA repair mechanisms of cancer cells, potentially improving treatment outcomes. The Company is conducting multiple clinical trials for solid tumors with unmet medical needs. LIXTE's unique approach has no known competitors and is covered by a comprehensive patent portfolio.
Forward-Looking Statements
This article was prepared by Hawk Point Media Group, LLC and may contain information, views, or opinions regarding the future expectations, plans, and prospects of Lixte Biotechnology Holdings, Inc. that constitute or may constitute forward-looking statements. These statements are not historical facts and are based on assumptions, beliefs, and expectations regarding future economic and operating performance. Although Hawk Point Media Group, LLC believes such statements are made in good faith and based on information available at the time of writing, there can be no assurance that the expectations expressed will prove accurate. Lixte Biotechnology Holdings, Inc. and Hawk Point Media Group, LLC undertake no obligation to update or revise any forward-looking statements, except as required by applicable law.
Forward-looking statements are inherently subject to risks, uncertainties, and factors that could cause actual results to differ materially from those projected. Such factors include, but are not limited to, industry conditions, regulatory developments, economic trends, and risks identified in Lixte Biotechnology Holdings, Inc.'s filings with the Securities and Exchange Commission. Readers are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date of publication.
Accuracy & Disclosure Statement: Hawk Point Media Group, LLC (HPM) works on a retainer basis with IR Agency, Inc. to provide press release, editorial, digital media, and consulting services. Accordingly, IR Agency, Inc. may engage HPM to create content relating to multiple public companies during the term of its retainer. Up to five thousand dollars of the retainer may be allocated toward the creation and syndication of the content printed above. Because of this arrangement, this content should be considered sponsored content. The information contained herein is based on sources believed to be reliable, including publicly available filings, company disclosures, and direct website content, and is accurate to the best of our knowledge at the time of creation. This content is for informational purposes only and should not be construed as investment advice. At the time of creation, HPM does not own, buy, sell, or trade securities of the companies covered. Any reproduction or syndication of this content must include this statement. This disclosure is made in accordance with Section 17(b) of the Securities Act of 1933, the Federal Trade Commission's Endorsement Guides, and other applicable regulations governing sponsored investment content.
Contact:
SOURCE: Lixte Biotechnology Holdings, Inc.
View the original press release on ACCESS Newswire
G.Stevens--AMWN