
-
 US begins 'biggest ever' Philippines war games in thick of Mideast war
US begins 'biggest ever' Philippines war games in thick of Mideast war
-
Anxiety lingers in divided Kashmir a year after shooting attack

-
 Hit reality show helps rev up Japan's delinquent youth subculture
Hit reality show helps rev up Japan's delinquent youth subculture
-
Oil prices bounce back on Iran war escalation

-
 Residents return to ravaged homes months after Hong Kong fire
Residents return to ravaged homes months after Hong Kong fire
-
Australia's Green wins playoff for third LPGA LA Championship title

-
 Pakistan's military chief takes lead on US-Iran talks in diplomatic blitz
Pakistan's military chief takes lead on US-Iran talks in diplomatic blitz
-
Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
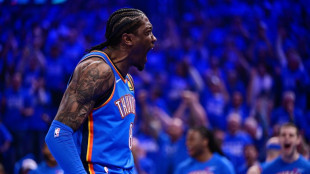
-
 US begins Philippines war games in thick of Middle East conflict
US begins Philippines war games in thick of Middle East conflict
-
Who's Bad? Not Michael Jackson in new big-budget biopic

-
 Nations gather for first-ever conference on fossil fuel exit
Nations gather for first-ever conference on fossil fuel exit
-
Money, lobbyists, inertia: why fossil fuels are so hard to quit

-
 France summons Elon Musk over X probe
France summons Elon Musk over X probe
-
'Save humanity': Four figures battling it out to lead embattled UN

-
 Gilgeous-Alexander, Wemby, Jokic finalists for NBA MVP
Gilgeous-Alexander, Wemby, Jokic finalists for NBA MVP
-
Israel vows to level homes in Lebanon, counter threats with 'full force'

-
 Ameriwest Completes 3D Geological Model Defining High-Grade Copper Shell Along with Two Additional Zones at the Bornite Project
Ameriwest Completes 3D Geological Model Defining High-Grade Copper Shell Along with Two Additional Zones at the Bornite Project
-
Genflow Biosciences PLC Announces Strategic Technology Collaboration

-
 QNX and NVIDIA Deepen Collaboration to Advance Safety-Critical Edge AI Across Robotics, Medical, and Industrial Systems
QNX and NVIDIA Deepen Collaboration to Advance Safety-Critical Edge AI Across Robotics, Medical, and Industrial Systems
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 20

-
 401(k) to Gold IRA Rollover Guide Released (2026 Update)
401(k) to Gold IRA Rollover Guide Released (2026 Update)
-
Who Does the Best Deep Plane Facelift in Florida?

-
 Rahm coasts to LIV Golf win in Mexico City
Rahm coasts to LIV Golf win in Mexico City
-
Fitzpatrick survives Scheffler playoff to win RBC Heritage

-
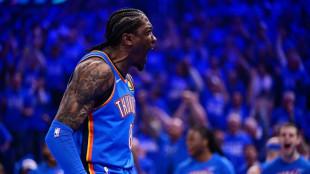 Thunder thrash Suns, Celtics crush Sixers in NBA playoff openers
Thunder thrash Suns, Celtics crush Sixers in NBA playoff openers
-
Bulgaria's former president tops parliamentary vote

-
 Kenyans Korir, Lokedi seek to repeat at Boston Marathon
Kenyans Korir, Lokedi seek to repeat at Boston Marathon
-
AC Milan, Juventus close in on Champions League qualification

-
 Spring double keeps Racing 92 in Top 14 play-off hunt with Paris derby win
Spring double keeps Racing 92 in Top 14 play-off hunt with Paris derby win
-
Endrick stars as Lyon dent PSG's Ligue 1 title hopes

-
 History haunts Arsenal as Man City take control of title race
History haunts Arsenal as Man City take control of title race
-
AC Milan and Juventus close in on Champions League qualification

-
 Celtics crush Sixers as Tatum and Brown shine in playoff opener
Celtics crush Sixers as Tatum and Brown shine in playoff opener
-
Guardiola warns title not won yet as Man City hunt down Arsenal

-
 Arteta tells Arsenal to 'go again' in pursuit of Premier League title
Arteta tells Arsenal to 'go again' in pursuit of Premier League title
-
Treble-chasing Bayern put beer showers on ice despite title win

-
 Eight children dead in US domestic violence shooting
Eight children dead in US domestic violence shooting
-
Arya, Connolly help Punjab hammer Lucknow in IPL

-
 Man City beat Arsenal to seize control of title race, Liverpool win
Man City beat Arsenal to seize control of title race, Liverpool win
-
Kane scores as Bayern sink Stuttgart to claim Bundesliga title

-
 Balogun continues Monaco scoring streak, Rennes boost Champions League hopes
Balogun continues Monaco scoring streak, Rennes boost Champions League hopes
-
Trump orders negotiators to Pakistan, but Iran on the fence over talks

-
 Haaland gives Man City edge over Arsenal in Premier League title showdown
Haaland gives Man City edge over Arsenal in Premier League title showdown
-
Slot hails Liverpool mentality after last-gasp derby winner

-
 Top boss vows 'no sitting still' as rugby bids to conquer US
Top boss vows 'no sitting still' as rugby bids to conquer US
-
Fils wins on Barcelona clay with French Open looming

-
 'Super Mario Galaxy' rules N. America box office for third week
'Super Mario Galaxy' rules N. America box office for third week
-
Liverpool snatch derby win ahead of City-Arsenal showdown

-
 Evenepoel outsprints Skjelmose to win Amstel Gold Race
Evenepoel outsprints Skjelmose to win Amstel Gold Race
-
Liverpool beat Everton ahead of City-Arsenal showdown


ClearPoint Neuro Announces the Appointment of Dr. Paul Larson as Chief Medical Officer
SOLANA BEACH, CALIFORNIA / ACCESS Newswire / September 25, 2025 / ClearPoint Neuro, Inc. (Nasdaq:CLPT) (the "Company"), a global device, cell, and gene therapy-enabling company offering precise navigation to the brain and spine, today announced that Dr. Paul Larson will be joining the company as Chief Medical Officer starting in January 2026. Dr. Larson has a long history of working with ClearPoint Neuro as an advisor, especially supporting education and product development efforts related to cell and gene therapy delivery. Dr. Larson will continue his appointments at both the University of California San Francisco and at Banner Hospital, University of Arizona where he will continue to enroll patients in various ongoing cell and gene therapy clinical trials.
"We are thrilled to have Paul join our team in this very significant role and at the perfect time," commented Joe Burnett, President and CEO of ClearPoint Neuro. "Paul has been a part of the ClearPoint Neuro history from the very start, through his expert knowledge of neuro navigation and drug delivery, as well as his passion for mentoring and coaching in totally new treatment paradigms. In the field of neuro cell and gene therapy, Paul has played an essential role alongside some of the earliest investigators in this space having personally performed or overseen more than one hundred cell and gene therapy clinical procedures. Now that ClearPoint Neuro and our partners are preparing for the commercialization of these new-to-world therapies, Paul is a perfect fit to both educate and amplify access to these therapies using his substantial knowledge of best practices for neuro drug delivery worldwide."
"This is a very exciting time for the field of neurosurgery as multiple new cell and gene therapy treatments are quickly progressing under the FDA expedited review process," commented Paul Larson, M.D. "These new direct-delivery drug platforms are crucially important if we as a medical profession want to make progress in not only treating symptoms, but in potentially altering the course of the underlying disease for disorders like Huntington's Disease, Parkinsons' Disease, epilepsy, dementias, tumors, rare childhood genetic disorders, stroke rehabilitation and more. Joining ClearPoint Neuro is a truly unique opportunity to continue my work advising many different partners across these diverse indications because the ClearPoint drug delivery ecosystem will be essential to many of these platforms. I do not have to pick a favorite but rather celebrate the progress across all of ClearPoint Neuro's sixty-plus partners."
About ClearPoint Neuro
ClearPoint Neuro is a device, cell, and gene therapy-enabling company offering precise navigation to the brain and spine. The Company uniquely provides both established clinical products as well as preclinical development services for controlled drug and device delivery. The Company's flagship product, the ClearPoint Neuro Navigation System, has FDA clearance and is CE-marked. ClearPoint Neuro is engaged with healthcare and research centers in North America, Europe, Asia, and South America. The Company is also partnered with the most innovative pharmaceutical/biotech companies, academic centers, and contract research organizations, providing solutions for direct central nervous system delivery of therapeutics in preclinical studies and clinical trials worldwide. To date, thousands of procedures have been performed and supported by the Company's field-based clinical specialist team, which offers support and services to our customers and partners worldwide. For more information, please visit www.clearpointneuro.com.
Forward-Looking Statements
This press release contains forward-looking statements within the context of the federal securities laws, including statements regarding the prospects of the Company's gene and cell therapy partnerships, including the continuation of these partnerships, the continuation of current and potential expedited review programs, and expectations for such therapies' commercialization plans and market opportunity. These forward-looking statements are based on management's current expectations and are subject to the risks inherent in the business, which may cause the Company's actual results to differ materially from those expressed in or implied by forward-looking statements. Particular uncertainties and risks include those relating to: macroeconomic and inflationary conditions; regulatory and policy uncertainty; the introduction of or changes in tariffs, sanctions, or trade barriers; changes in monetary policy; geopolitical trends, such as protectionism and economic nationalism; future revenue from sales of the Company's products and services; the Company's ability to market, commercialize and achieve broader market acceptance for new products and services offered by the Company; the ability of our biologics and drug delivery partners to achieve commercial success, including their use of the Company's products and services in their delivery of therapies; the Company's ability to maintain its current relationships with biologics and drug delivery partners or enter into new relationships with such partners; the Company's expectations, projections and estimates regarding expenses, future revenue, capital requirements, and the availability of and the need for additional financing; the Company's ability to obtain additional funding to support its research and development programs; the ability of the Company to manage the growth of its business; the Company's ability to attract and retain its key employees; and risks inherent in the research, development, and regulatory approval of the Company's new products, including the ability to obtain and maintain regulatory clearances; manufacturing scale-up; supply chain and quality system readiness; intellectual property protection and freedom to operate; market acceptance and competitive dynamics. More detailed information on these and additional factors that could affect the Company's actual results are described in the "Risk Factors" section of the Company's Annual Report on Form 10-K for the year ended December 31, 2024, and the Company's Quarterly Report on Form 10-Q for the three months ended June 30, 2025, both of which have been filed with the Securities and Exchange Commission. The Company does not assume any obligation to update these forward-looking statements.
Contact:
Investor Relations:
Danilo D'Alessandro, Chief Financial Officer
(888) 287-9109 ext. 3
[email protected]
SOURCE: ClearPoint Neuro, Inc.
View the original press release on ACCESS Newswire
P.Martin--AMWN
