
-
 Anxiety lingers in divided Kashmir a year after shooting attack
Anxiety lingers in divided Kashmir a year after shooting attack
-
Hit reality show helps rev up Japan's delinquent youth subculture

-
 Oil prices bounce back on Iran war escalation
Oil prices bounce back on Iran war escalation
-
Residents return to ravaged homes months after Hong Kong fire

-
 Australia's Green wins playoff for third LPGA LA Championship title
Australia's Green wins playoff for third LPGA LA Championship title
-
Pakistan's military chief takes lead on US-Iran talks in diplomatic blitz

-
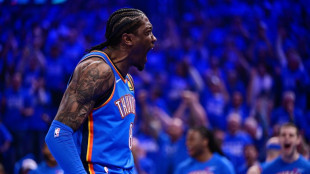 Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
-
US begins Philippines war games in thick of Middle East conflict

-
 Who's Bad? Not Michael Jackson in new big-budget biopic
Who's Bad? Not Michael Jackson in new big-budget biopic
-
Nations gather for first-ever conference on fossil fuel exit

-
 Money, lobbyists, inertia: why fossil fuels are so hard to quit
Money, lobbyists, inertia: why fossil fuels are so hard to quit
-
France summons Elon Musk over X probe

-
 'Save humanity': Four figures battling it out to lead embattled UN
'Save humanity': Four figures battling it out to lead embattled UN
-
Gilgeous-Alexander, Wemby, Jokic finalists for NBA MVP

-
 Israel vows to level homes in Lebanon, counter threats with 'full force'
Israel vows to level homes in Lebanon, counter threats with 'full force'
-
401(k) to Gold IRA Rollover Guide Released (2026 Update)

-
 Who Does the Best Deep Plane Facelift in Florida?
Who Does the Best Deep Plane Facelift in Florida?
-
Rahm coasts to LIV Golf win in Mexico City

-
 Fitzpatrick survives Scheffler playoff to win RBC Heritage
Fitzpatrick survives Scheffler playoff to win RBC Heritage
-
Thunder thrash Suns, Celtics crush Sixers in NBA playoff openers
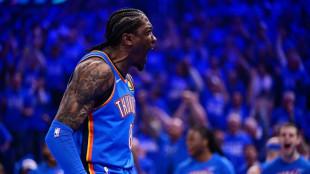
-
 Bulgaria's former president tops parliamentary vote
Bulgaria's former president tops parliamentary vote
-
Kenyans Korir, Lokedi seek to repeat at Boston Marathon

-
 AC Milan, Juventus close in on Champions League qualification
AC Milan, Juventus close in on Champions League qualification
-
Spring double keeps Racing 92 in Top 14 play-off hunt with Paris derby win

-
 Endrick stars as Lyon dent PSG's Ligue 1 title hopes
Endrick stars as Lyon dent PSG's Ligue 1 title hopes
-
History haunts Arsenal as Man City take control of title race

-
 AC Milan and Juventus close in on Champions League qualification
AC Milan and Juventus close in on Champions League qualification
-
Celtics crush Sixers as Tatum and Brown shine in playoff opener

-
 Guardiola warns title not won yet as Man City hunt down Arsenal
Guardiola warns title not won yet as Man City hunt down Arsenal
-
Arteta tells Arsenal to 'go again' in pursuit of Premier League title

-
 Treble-chasing Bayern put beer showers on ice despite title win
Treble-chasing Bayern put beer showers on ice despite title win
-
Eight children dead in US domestic violence shooting

-
 Arya, Connolly help Punjab hammer Lucknow in IPL
Arya, Connolly help Punjab hammer Lucknow in IPL
-
Man City beat Arsenal to seize control of title race, Liverpool win

-
 Kane scores as Bayern sink Stuttgart to claim Bundesliga title
Kane scores as Bayern sink Stuttgart to claim Bundesliga title
-
Balogun continues Monaco scoring streak, Rennes boost Champions League hopes

-
 Trump orders negotiators to Pakistan, but Iran on the fence over talks
Trump orders negotiators to Pakistan, but Iran on the fence over talks
-
Haaland gives Man City edge over Arsenal in Premier League title showdown

-
 Slot hails Liverpool mentality after last-gasp derby winner
Slot hails Liverpool mentality after last-gasp derby winner
-
Top boss vows 'no sitting still' as rugby bids to conquer US

-
 Fils wins on Barcelona clay with French Open looming
Fils wins on Barcelona clay with French Open looming
-
'Super Mario Galaxy' rules N. America box office for third week

-
 Liverpool snatch derby win ahead of City-Arsenal showdown
Liverpool snatch derby win ahead of City-Arsenal showdown
-
Evenepoel outsprints Skjelmose to win Amstel Gold Race

-
 Liverpool beat Everton ahead of City-Arsenal showdown
Liverpool beat Everton ahead of City-Arsenal showdown
-
Rabiot fires AC Milan past Verona to verge of Champions League return

-
 UK PM vows to find arsonists of London Jewish sites
UK PM vows to find arsonists of London Jewish sites
-
Rinku blitz leads Kolkata to first win of IPL season

-
 Shelton wins fifth ATP title with victory in Munich
Shelton wins fifth ATP title with victory in Munich
-
UK's Starmer to face grilling from MPs over Mandelson scandal


IGC Pharma Adds Ichor Research as New Site for CALMA Phase 2 Alzheimer's Trial
POTOMAC, MD / ACCESS Newswire / September 29, 2025 / IGC Pharma, Inc. (NYSE American:IGC) today announced the addition of a new clinical trial site to its ongoing Phase 2 study evaluating IGC-AD1, an investigational drug candidate for treating agitation in Alzheimer's disease. The new site, Ichor Research, located in Syracuse, New York, is led by Principal Investigator Dr. Karl F. Hafner, M.D.

Dr. Hafner brings years of experience in clinical research and patient care, particularly in the field of neurodegenerative conditions. Ichor Research began enrolling eligible participants for the CALMA trial immediately.
The CALMA Phase 2 trial is a randomized, double-blind, placebo-controlled study designed to evaluate the safety and efficacy of IGC-AD1 in reducing agitation symptoms in individuals living with Alzheimer's disease. Agitation is one of the most prevalent and distressing behavioral symptoms in Alzheimer's patients, often accelerating disease progression and increasing caregiver burden.
"We are proud to partner with Ichor Research as part of our commitment to expand access to innovative treatment options for patients and caregivers," said Ram Mukunda, CEO of IGC Pharma. "Dr. Hafner, and the team at Ichor bring valuable expertise that will help advance the CALMA trial."
Contact Information:
Kristen Stebbins
VP of Clinical Trial Operations
1101 Erie Blvd East, Suite 201
Syracuse, NY 13210
Office: 315-904-5004
Email: [email protected]
To learn more about IGC Pharma and the CALMA trial, please visit: Link
About Ichor Research
Research Central LLC, operating as Ichor Research, is a clinical research organization based in Syracuse, New York, with expertise in clinical trials for Alzheimer's disease. Ichor Research is dedicated to overcoming the unique challenges involved in Alzheimer's disease clinical trials, advancing medical innovation through high-quality, patient-focused clinical trials. Under the leadership of experienced clinical trials experts Lisa Sonneborn and Kristen Stebbins, Ichor collaborates with industry sponsors to evaluate novel treatments across a variety of therapeutic areas. The site is committed to ethical research practices, scientific integrity, and improving outcomes for patients through the development of evidence-based medical therapies.
About IGC Pharma (dba IGC):
IGC Pharma (NYSE American: IGC) is a clinical-stage biotechnology company leveraging AI to develop innovative treatments for Alzheimer's and metabolic disorders. Our lead asset, IGC-AD1, is a cannabinoid-based therapy currently in a Phase 2 trial (CALMA) for agitation in Alzheimer's dementia. Our pipeline includes TGR-63, targeting amyloid plaques, and early-stage programs focused on neurodegeneration, tau proteins, and metabolic dysfunctions. We integrate AI to accelerate drug discovery, optimize clinical trials, and enhance patient targeting. With 30 patent filings and a commitment to innovation, IGC Pharma is advancing breakthrough therapies.
Forward-Looking Statements:
This press release contains forward-looking statements. These forward-looking statements are based largely on IGC Pharma's expectations and are subject to several risks and uncertainties, certain of which are beyond IGC Pharma's control. Actual results could differ materially from these forward-looking statements as a result of, among other factors, the Company's failure or inability to commercialize one or more of the Company's products or technologies, including the products or formulations described in this release, or failure to obtain regulatory approval for the products or formulations, where required, or government regulations affecting AI or the AI algorithms not working as intended or producing accurate predictions; general economic conditions that are less favorable than expected; the FDA's general position regarding cannabis- and hemp-based products; and other factors, many of which are discussed in IGC Pharma's U.S. Securities and Exchange Commission ("SEC") filings. IGC incorporates by reference its Annual Report on Form 10-K filed with the SEC on June 27, 2025, as if fully incorporated and restated herein. Considering these risks and uncertainties, there can be no assurance that the forward-looking information contained in this release will occur. IGC Pharma, Inc. assumes no obligation to update forward-looking statements contained in this release as the result of new information or future events or developments.
Contact Information
Rosalyn Christian / John Nesbett
IMS Investor Relations
[email protected]
(203) 972-9200
SOURCE: IGC Pharma, Inc.
View the original press release on ACCESS Newswire
J.Oliveira--AMWN

