-
 Nuggets hold off T'Wolves, Cavs thump Raptors in NBA playoff openers
Nuggets hold off T'Wolves, Cavs thump Raptors in NBA playoff openers
-
Fitzpatrick extends lead as Scheffler charges at RBC Heritage

-
 Real Sociedad secure Copa del Rey penalty triumph over Atletico
Real Sociedad secure Copa del Rey penalty triumph over Atletico
-
'Scandalous' Marseille lose at Lorient, dent Champions League bid

-
 Arteta urges Arsenal to have no regrets in Man City title showdown
Arteta urges Arsenal to have no regrets in Man City title showdown
-
Substitute Dupont helps Toulouse cruise past Castres in Top 14

-
 Questions surround Warriors after NBA play-in exit
Questions surround Warriors after NBA play-in exit
-
Man Utd beat Chelsea as Spurs stunned by Brighton equaliser

-
 Cunha steers Man Utd towards Champions League at Chelsea's expense
Cunha steers Man Utd towards Champions League at Chelsea's expense
-
Cavs cruise past Raptors in NBA playoff opener

-
 England beat Iceland to stay perfect in Women's World Cup qualifying
England beat Iceland to stay perfect in Women's World Cup qualifying
-
Spurs 'not finished yet', says defiant De Zerbi

-
 Germany's Gnabry a World Cup doubt after thigh injury
Germany's Gnabry a World Cup doubt after thigh injury
-
Spurs stunned by late Brighton equaliser, Leeds pull clear of trouble

-
 Spurs count cost after Brighton draw leaves them in drop zone
Spurs count cost after Brighton draw leaves them in drop zone
-
'Scandalous' Marseille lose at Lorient, damage Champions League bid

-
 Abhishek fireworks, Malinga spell sink Chennai
Abhishek fireworks, Malinga spell sink Chennai
-
Napoli's Serie A title defence nears end with Lazio defeat

-
 England run in 12 tries to hammer Scotland in Six Nations
England run in 12 tries to hammer Scotland in Six Nations
-
Rybakina powers past Andreeva to reach Stuttgart final

-
 At least 5 killed after gunman opens fire in Ukrainian capital
At least 5 killed after gunman opens fire in Ukrainian capital
-
Bayern on cusp of title as Dortmund lose, Eta beaten on debut

-
 Rublev, Fils fightbacks set up Barcelona Open final
Rublev, Fils fightbacks set up Barcelona Open final
-
Leeds pull clear of trouble, Bournemouth sink Newcastle

-
 Spain rout Ukraine to boost Women's World Cup qualifying hopes
Spain rout Ukraine to boost Women's World Cup qualifying hopes
-
Bayern close in on Bundesliga title as Dortmund lose

-
 Iran closes Hormuz Strait again, as Trump warns against 'blackmail'
Iran closes Hormuz Strait again, as Trump warns against 'blackmail'
-
US extends sanctions waiver on purchases of Russian oil

-
 Trump signs order to fast-track research on psychedelic drugs
Trump signs order to fast-track research on psychedelic drugs
-
Cobolli downs Zverev to set up Munich final with Shelton

-
 Pope arrives in Angola on Africa tour overshadowed by Trump
Pope arrives in Angola on Africa tour overshadowed by Trump
-
Thousands protest in Germany urging faster green shift

-
 La Rochelle thump threadbare Bordeaux-Begles
La Rochelle thump threadbare Bordeaux-Begles
-
Muchova battles past Svitolina to book Stuttgart final berth

-
 Allegri rules out taking Italy job, wants to stay at AC Milan
Allegri rules out taking Italy job, wants to stay at AC Milan
-
Miller bludgeons Delhi to IPL win over Bengaluru

-
 Pope says he regrets his remarks interpreted as a debate with Trump
Pope says he regrets his remarks interpreted as a debate with Trump
-
Brentford blow chance for top six in Fulham stalemate

-
 Trade ships hit in Hormuz as Iran reopening falters
Trade ships hit in Hormuz as Iran reopening falters
-
France blames Hezbollah for French peacekeeper's death in Lebanon

-
 Venezuela's Machado doesn't regret gifting Nobel Peace Prize to Trump
Venezuela's Machado doesn't regret gifting Nobel Peace Prize to Trump
-
No date set for next round of Iran-US talks: Iran deputy FM

-
 Iran closes Hormuz Strait again over US blockade, ships reverse course
Iran closes Hormuz Strait again over US blockade, ships reverse course
-
'We've already beaten other favorites', Lyon's Endrick warns PSG

-
 Turkey says Israel using security as a pretext to acquire 'more land'
Turkey says Israel using security as a pretext to acquire 'more land'
-
Iran closes Hormuz Strait again over US blockade with ships mid-transit

-
 French film star Nathalie Baye dead at 77: family to AFP
French film star Nathalie Baye dead at 77: family to AFP
-
China sex toy makers cautiously embrace AI wave

-
 Paramount's CinemaCon charm offensive gets lukewarm reception
Paramount's CinemaCon charm offensive gets lukewarm reception
-
Game over: Players press EU to ban 'destroying' video titles


C. Light Named One of Five Selected Projects in the Roche MS Innovation Challenge
Two-Year Clinical Trial with UCSF and Sutter Health Will Study Fixational Eye Movements as a Biomarker of MS Progression
SANTA CLARA, CA / ACCESS Newswire / November 19, 2025 / C. Light Technologies, an AI-driven healthtech company specializing in assessing eye and brain health to aid in early detection and earlier intervention of neurodegenerative diseases, has been selected as one of five project teams that will be funded through the Roche MS Innovation Challenge. C. Light will use the funding to launch a two-year, multi-site clinical trial, partnering with leading multiple sclerosis (MS) centers at UCSF and Sutter Health, Palo Alto, Calif. to identify hard-to-detect, fixational eye movement outputs as non-invasive biomarkers for MS progression.
The Roche MS Innovation Challenge is a global initiative designed to accelerate breakthroughs that improve the lives of people with MS. It aims to bring forward practical innovations that could be implemented in clinical care within the next few years. The research trial will use C. Light's proprietary Retitrack™ scanning laser ophthalmoscopy (SLO) technology-the first FDA 510k-cleared retinal eye-movement monitor-to record eye movements at five timepoints over the course of two years.
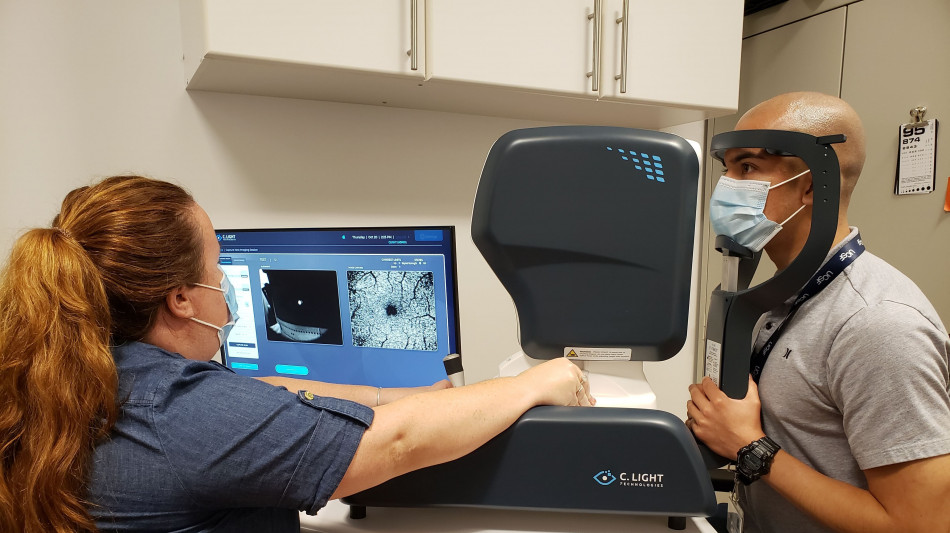
The Retitrack scanning laser ophthalmoscopy (SLO) technology
The study will aim to correlate changes in microscopic eye movements with subtle signs of PIRA (progression independent of relapse activity), including individuals already treating their condition with B-cell depleting therapies. It aims to determine if Retitrack™-detected fixational and saccadic eye-motion changes will predict PIRA earlier than the Expanded Disability Status Scale (EDSS)-the widely accepted standardized measure providing a quantitative evaluation of MS disability level-and MRI.
C. Light has shown in earlier research studies that fixational eye movements change with MS and correlate with EDSS progression. The Retitrack™ captures one of the smallest motor movements the human body can make-fixation; tiny, involuntary eye movements that can be smaller than half the size of a human hair. It measures this movement with a 10-second, non-invasive eye scan, requiring no eye dilation.
"We are creating an entirely new stream of data for healthcare," said C. Light Founder and CEO Christy Sheehy-Bensinger. "Why wait until ambulation and mobility are already impacted in a patient to document disease progression and disability? Let's use the subclinical changes of oculomotor function and fixation to detect subtle MS progression as soon as it occurs."
In July, the American Medical Association (AMA) clinical board granted C. Light a new CPT code (1010T) for retinal eye tracking, which will become effective for clinical use on Jan. 1, 2026. This opens the door for potential insurance reimbursement for Retitrack scans.
C. Light's Chief Medical Officer Jacqueline Theis, O.D., F.A.A.O. said, "Retitrack's ability to quantify microscopic fixational eye-movement changes offers a new lens on PIRA and MS detection -one that could shorten trial endpoints, refine therapeutic evaluation, and enable more personalized patient monitoring. This is the kind of bench-to-bedside innovation that has the power to transform both the science of drug development and the lived experience of MS."
In May, C. Light closed an oversubscribed $3 million seed extension round led by a large North American family office to fuel their upcoming FDA clinical trial to add AI-powered disease detection capabilities to their device.
C. Light is also announcing its participation in the DayOne Accelerator in Basel, Switzerland, a three-month, hybrid program that prepares startups to launch successful partnerships with pharmaceutical companies. Dr. Sheehy will be attending their Innovation Showcase Dec. 10 and 11 at the Roche campus in Basel, Switzerland.
About C. Light
C. Light Technologies, Inc. is an AI-driven healthtech company whose primary mission is to create digital tools and solutions to assess eye and brain health. Its proprietary Retitrack™ device is reshaping the landscape of eye-tracking, enabling clinicians to unlock the untapped potential within each eye movement. C. Light has raised more than $8 million in funding with major investors Yamaha Motor Ventures and Creative Ventures. The company has been awarded three grants from the National Institute of Health, as well as additional funding through the Alzheimer's Drug Discovery Foundation. C. Light has also won numerous awards in recent years, including the 2022 National Timmy Award for "Best Tech Startup in North America" and the 2023 Stevie® Award for Most Innovative Company of the Year - 10 or Less Employees.
For more information, visit our website at www.clighttechnologies.com or our LinkedIn.
Disclaimer: The information described above is conducted as research through controlled studies and is not intended to reflect the ability of the current commercially available iteration of the Retitrack.
Images: LINK
CONTACT:
Erica Zeidenberg
+19255188159
[email protected]
SOURCE: C. Light Technologies
View the original press release on ACCESS Newswire
A.Rodriguezv--AMWN