
-
 France's leftists win mayoral elections in largest cities
France's leftists win mayoral elections in largest cities
-
Asian stocks tumble as Trump gives Iran 48-hour ultimatum

-
 Wolves rally past Celtics, Nuggets sink Blazers
Wolves rally past Celtics, Nuggets sink Blazers
-
Middle East war to dominate Houston's 'Davos of Energy'

-
 Kim holds off Korda charge to win LPGA Founders Cup
Kim holds off Korda charge to win LPGA Founders Cup
-
Trump orders immigration agents to airports amid crippling budget standoff

-
 Iran awaits Trump threat to blow up power plants
Iran awaits Trump threat to blow up power plants
-
Alcaraz eyes clay court season after early Miami exit

-
 Real Madrid down Atletico in derby, leaders Barca edge Rayo
Real Madrid down Atletico in derby, leaders Barca edge Rayo
-
Korda sends Alcaraz to another early exit in Miami

-
 Bordeaux-Begles hammer Toulouse in Dupont absence
Bordeaux-Begles hammer Toulouse in Dupont absence
-
Slovenia PM claims election win as results show neck and neck finish

-
 England's Fitzpatrick birdies 18th to win PGA Valspar title
England's Fitzpatrick birdies 18th to win PGA Valspar title
-
Man City's League Cup glory adds twist to title race

-
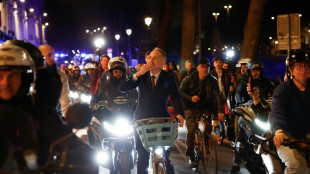 Leftists win mayoral elections in Paris and Marseille
Leftists win mayoral elections in Paris and Marseille
-
Vinicius double helps Real Madrid edge Atletico thriller

-
 Doncic cleared to face Pistons after foul rescinded: NBA
Doncic cleared to face Pistons after foul rescinded: NBA
-
Inter's Serie A lead cut to six with Fiorentina draw, Como march on

-
 World No.1 Alcaraz beaten by Korda in Miami Open third round
World No.1 Alcaraz beaten by Korda in Miami Open third round
-
Cuba starts to restore power after new blackout

-
 Ovechkin nets 1,000th combined NHL season-playoffs goal
Ovechkin nets 1,000th combined NHL season-playoffs goal
-
Undav doubles up as Stuttgart down Augsburg to go third

-
 Leftists win mayoral elections in Paris and Marseille: projections
Leftists win mayoral elections in Paris and Marseille: projections
-
Israel warns weeks of fighting ahead in Mideast war

-
 Guardiola revels in Man City's 'special' League Cup win over Arsenal
Guardiola revels in Man City's 'special' League Cup win over Arsenal
-
Hodgkinson headlines Britain's 'Super Sunday' at world indoors

-
 Messi scores for Miami in 3-2 MLS victory at NYCFC
Messi scores for Miami in 3-2 MLS victory at NYCFC
-
Bezzecchi wins second race of the season at Brazil MotoGP

-
 Britain's Hodgkinson wins world indoor 800m gold
Britain's Hodgkinson wins world indoor 800m gold
-
Former France and West Ham star Payet announces retirement

-
 Man City's O'Reilly savours 'unbelievable' double in League Cup final win
Man City's O'Reilly savours 'unbelievable' double in League Cup final win
-
Israel to advance ground operations in Lebanon after striking key bridge

-
 Man City win League Cup as O'Reilly sinks Arsenal after Kepa blunder
Man City win League Cup as O'Reilly sinks Arsenal after Kepa blunder
-
Marseille downed by Lille in Ligue 1 as Lyon's struggles continue

-
 NBA bans Mitchell, Champagnie one game for sparking melee
NBA bans Mitchell, Champagnie one game for sparking melee
-
'Project Hail Mary' rockets to top of N. America box office

-
 Syrians protest alcohol sale limits, curbs on personal freedom
Syrians protest alcohol sale limits, curbs on personal freedom
-
Spurs can '100 percent' avoid nightmare of relegation: Saltor

-
 Araujo header scrapes Liga leaders Barcelona win over Rayo
Araujo header scrapes Liga leaders Barcelona win over Rayo
-
Israel launches strikes as Lebanon warns of invasion

-
 Torrential rains in Kenya kill 81 in March: officials
Torrential rains in Kenya kill 81 in March: officials
-
Iran threatens Mideast infrastructure after Trump ultimatum

-
 Spurs felled by Forest in relegation battle, Sunderland shock Newcastle
Spurs felled by Forest in relegation battle, Sunderland shock Newcastle
-
Spurs collapse against Forest, failing acid test

-
 US may 'escalate to de-escalate' against Iran: Treasury chief
US may 'escalate to de-escalate' against Iran: Treasury chief
-
Howe disappointed in himself after 'painful' Newcastle defeat

-
 Quansah to miss England's pre-World Cup friendlies
Quansah to miss England's pre-World Cup friendlies
-
Araujo header scrapes Liga leaders Barca win over Rayo

-
 Georgia buries Patriarch Ilia II as succession stirs fears of Russian influence
Georgia buries Patriarch Ilia II as succession stirs fears of Russian influence
-
DeChambeau wins back-to-back LIV Golf play-offs


Moderna Announces Phase 3 Study of Investigational Cytomegalovirus (CMV) Vaccine Did Not Meet Primary Efficacy Endpoint
Company to discontinue development of mRNA-1647 in congenital CMV
Company will continue to evaluate mRNA-1647 in ongoing Phase 2 trial in bone marrow transplant patients
Company does not anticipate any impact to its 2025 financial guidance or its expectation of achieving breakeven in 2028
CAMBRIDGE, MA / ACCESS Newswire / October 22, 2025 / Moderna, Inc. (NASDAQ:MRNA) today announced topline results from a Phase 3 pivotal trial evaluating the efficacy of mRNA-1647, the Company's investigational cytomegalovirus (CMV) vaccine. The study did not meet its primary efficacy endpoint of preventing CMV infection in seronegative female participants of childbearing age (16-40 years of age). Moderna will discontinue its congenital CMV clinical development program.
"Today's announcement is disappointing for families and healthcare professionals who have been eagerly awaiting a CMV vaccine to prevent congenital CMV, a leading infectious cause of birth defects," said Stéphane Bancel, Chief Executive Officer of Moderna. "The majority of the data from this trial is still forthcoming, and we will share our full results with the scientific community in hopes that our learnings can contribute to the continued pursuit of a CMV vaccine. I am grateful for our dedicated team, our partners at clinical trial sites, and all the trial participants who made this research possible."
The Phase 3 trial (NCT05085366) was a randomized, observer-blind, placebo-controlled study. The study included approximately 7,500 women 16-40 years of age from approximately 300 sites across 13 countries, representing the largest trial assessing the efficacy of a CMV vaccine to-date. Vaccine efficacy against primary CMV infection in healthy women was well below the Company's target, ranging from 6% to 23% depending on the case definition used.
"We are clearly disappointed by the failure to prevent primary infection because it means there is still no vaccine for the prevention of congenital CMV despite the many decades of work by the field," said Stephen Hoge, MD, President of Moderna. "CMV does cause significant disease in other contexts, including reactivation of the latent virus in those undergoing bone marrow transplantation, and we will continue to explore the potential of mRNA-1647 to suppress disease associated with reactivation in those high-risk patients through our ongoing Phase 2 study."
mRNA-1647 was found to be generally well-tolerated regardless of CMV serostatus, with a safety profile consistent with earlier studies. No safety concerns have been raised by the Data and Safety Monitoring Board. The Company plans to continue its study of mRNA-1647 in bone marrow transplant patients, which is currently in a Phase 2 trial (NCT05683457).
Moderna does not anticipate any impact to its 2025 financial guidance or its expectation of achieving breakeven in 2028. Moderna anticipated minimal initial revenue contribution from mRNA-1647 given necessary investments in market building and launch, and expected the product to be cash-flow negative in 2028.
About Moderna
Moderna is a leader in the creation of the field of mRNA medicine. Through the advancement of mRNA technology, Moderna is reimagining how medicines are made and transforming how we treat and prevent disease for everyone. By working at the intersection of science, technology and health for more than a decade, the company has developed medicines at unprecedented speed and efficiency, including pioneering work on COVID-19 vaccines.
Moderna's mRNA platform has enabled the development of therapeutics and vaccines for infectious diseases, immuno-oncology, rare diseases and autoimmune diseases. With a unique culture and a global team driven by the Moderna values and mindsets to responsibly change the future of human health, Moderna strives to deliver the greatest possible impact to people through mRNA medicines. For more information about Moderna, please visit modernatx.com and connect with us on X (formerly Twitter), Facebook, Instagram, YouTube and LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including statements regarding: Moderna's 2025 financial guidance and its expectation of achieving breakeven in 2028; and the potential of mRNA-1647 for bone marrow transplant patients and Moderna's ongoing Phase 2 trial. The forward-looking statements in this press release are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond Moderna's control and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks, uncertainties, and other factors include, among others, those risks and uncertainties described under the heading "Risk Factors" in Moderna's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, and in subsequent filings made by Moderna with the U.S. Securities and Exchange Commission, which are available on the SEC's website at www.sec.gov. Except as required by law, Moderna disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release in the event of new information, future developments or otherwise. These forward-looking statements are based on Moderna's current expectations and speak only as of the date of this press release.
Moderna Contacts
Media:
Chris Ridley
Head of Global Media Relations
+1 617-800-3651
[email protected]
Investors:
Lavina Talukdar
Senior Vice President & Head of Investor Relations
+1 617-209-5834
[email protected]
SOURCE: Moderna, Inc.
View the original press release on ACCESS Newswire
Ch.Kahalev--AMWN

