
-
 French elect mayors in key cities including Paris
French elect mayors in key cities including Paris
-
'They beat us with whips': Sudan RSF detainees tell of horrors in El-Fasher

-
 Australia's Hannah Green wins historic third tournament in a row
Australia's Hannah Green wins historic third tournament in a row
-
China's premier vows to expand global 'trade pie': state media

-
 Belgium commemorates Brussels attacks 10 years on
Belgium commemorates Brussels attacks 10 years on
-
Sri Lanka raises fuel prices by 25 percent as war bites

-
 Rights groups fear use of arrest to stifle free speech in Pakistan
Rights groups fear use of arrest to stifle free speech in Pakistan
-
Iranian missiles sow panic, destruction in Israeli towns

-
 Damaged Russian tanker to be towed to Libya: state-owned company
Damaged Russian tanker to be towed to Libya: state-owned company
-
Gilgeous-Alexander scores 40, LeBron breaks NBA appearance record

-
 Cuba hit by second nationwide blackout in a week
Cuba hit by second nationwide blackout in a week
-
BTS draws over 100,000 fans to Seoul comeback concert: label

-
 US-China 'Board of Trade' may help ties but experts flag market worries
US-China 'Board of Trade' may help ties but experts flag market worries
-
Sinner, defending champ Mensik advance to third round at Miami Open

-
 Iran missile strikes wound over 100 in two south Israel towns
Iran missile strikes wound over 100 in two south Israel towns
-
Shai hits 40 as Thunder win despite NBA melee with four ejected

-
 Records shattered as US heatwave moves eastward
Records shattered as US heatwave moves eastward
-
Iran missiles hit southern Israel, injuring more than 100

-
 LeBron James breaks record for most NBA games played
LeBron James breaks record for most NBA games played
-
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Japan coach says Asian Cup crown 'well-deserved' for inspirational team
Japan coach says Asian Cup crown 'well-deserved' for inspirational team
-
PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
-
Milan move to within five points of Serie A leaders Inter

-
 Duplantis masterclass as Kerr and record-setter Ehammer shine
Duplantis masterclass as Kerr and record-setter Ehammer shine
-
Rosenior urges Chelsea to 'forget the noise' after damaging loss

-
 Marquez ambushed Di Giannantonio to win Brazil sprint
Marquez ambushed Di Giannantonio to win Brazil sprint
-
Sweden's Duplantis wins fourth world indoor pole vault title

-
 Liverpool, Chelsea slip up in Champions League race
Liverpool, Chelsea slip up in Champions League race
-
WHO sends first overland convoy from emergencies hub to Beirut

-
 Everton rub salt in Chelsea wounds as Champions League race tightens
Everton rub salt in Chelsea wounds as Champions League race tightens
-
Coach Mignoni returns but Toulon crash to Stade Francais

-
 Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
-
Sinner and Pegula advance to third round at Miami Open

-
 Britain's Kerr outsprints Hocker for world indoor 3,000m gold
Britain's Kerr outsprints Hocker for world indoor 3,000m gold
-
Kane backs Tuchel's call to rest him from England friendly

-
 NBA fines 76ers' Drummond, Magic's Suggs $25,000 each
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each
-
Switzerland's Ehammer sets indoor heptathlon world record

-
 Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set
-
World Athletics decision to hand Asia two world indoors 'strategic' - Coe

-
 Trump threatens to use ICE agents for airport security control
Trump threatens to use ICE agents for airport security control
-
Kane moves closer to goals record as Bayern sink Union

-
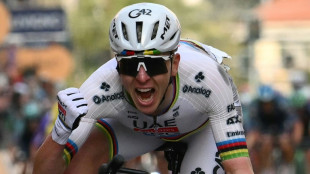 Pogacar ends long wait for Milan-San Remo glory after edging epic
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
US says 'took out' Iran base threatening blocked Hormuz oil route

-
 Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez
-
Welbeck scores twice to dent Liverpool's top-five hopes

-
 US strikes Iran bases threatening blocked Hormuz oil route
US strikes Iran bases threatening blocked Hormuz oil route
-
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin

-
 Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast
-
K-pop kings BTS stun Seoul in '2.0' comeback concert


Revelation Biosciences, Inc. Announces Financial Results for the Three and Nine Months Ended September 30, 2025
SAN DIEGO, CALIFORNIA / ACCESS Newswire / November 6, 2025 / Revelation Biosciences, Inc. (NASDAQ:REVB) (the "Company" or "Revelation"), a clinical-stage life sciences company that is focused on rebalancing inflammation to optimize health, today reported its financial results for the three and nine months ended September 30, 2025.
Corporate Highlights
Announced Groundbreaking Top-line Results from PRIME Clinical Study
Received gross proceeds of $9.6 million from warrant inducement in September 2025
"The outstanding PRIME data and subsequent financing validate Gemini's potential and places the company on solid footing to advance the next phase of development," said James Rolke, Chief Executive Officer of Revelation. "We look forward to meeting with the FDA later this year to gain agreement on the clinical development path to registration of Gemini, in turn enhancing shareholder value."
Results of Operations
As of September 30, 2025, Revelation had $12.7 million in cash and cash equivalents, compared to $6.5 million as of December 31, 2024. The increase in cash and cash equivalents was primarily due to net cash proceeds from the May 2025 public offering and the September 2025 warrant inducement, offset by cash used for operating activities. Based on current operating plans and projections, Revelation believes that its current cash and cash equivalents are sufficient to fund operations through the third quarter of 2026
Revelation's net cash used for operating activities for the nine months ended September 30, 2025 was $6.3 million compared to net cash used for operating activities of $14.6 million for the same period in 2024. Revelation's net loss for the three months ended September 30, 2025 was $1.9 million, or $(1.77) basic and diluted net loss per share compared to a net loss of $2.2 million, or $(40.15) basic and diluted net loss per share for the same period in 2024. Revelation's net loss for nine months ended September 30, 2025 was $6.4 million, or $(9.76) basic and diluted net loss per share compared to net loss of $13.3 million, or $(354.05) basic and diluted net loss per share for the same period in 2024.
About Gemini
Gemini is an intravenously administered, proprietary formulation of phosphorylated hexaacyl disaccharide (PHAD®) that reduces the damage associated with inflammation by reprogramming the innate immune system to respond to stress (trauma, infection, etc.) in an attenuated manner.
Gemini has the potential to treat a wide range of acute and chronic inflammatory conditions. Our primary focus is on the treatment of acute kidney injury (Gemini-AKI) as there are currently no therapies available to treat the large AKI market, and we believe this acute condition will provide the shortest pathway to marketing approval. In addition to AKI, Gemini is being evaluated for the treatment of chronic kidney disease (GEMINI-CKD program), post-burn infection and hyper-inflammatory response (Gemini-PBI) and post-surgical infection (GEMINI-PSI program).
The potential of Gemini has been demonstrated in multiple preclinical studies, previously announced positive Phase 1 clinical data, and in the recently announced positive Phase 1b clinical data in CKD patients.
About Revelation Biosciences, Inc.
Revelation Biosciences, Inc. is a clinical stage life sciences company focused on rebalancing inflammation using its proprietary formulation Gemini. Revelation has multiple ongoing programs to evaluate Gemini, including as a prevention for post-surgical infection, as prevention for acute kidney injury and for the treatment of chronic kidney disease.
For more information, please visit www.RevBiosciences.com.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These forward-looking statements are generally identified by the words "anticipate", "believe", "expect", "estimate", "plan", "outlook", and "project" and other similar expressions. We caution investors that forward-looking statements are based on management's expectations and are only predictions or statements of current expectations and involve known and unknown risks, uncertainties and other factors that may cause actual results to be materially different from those anticipated by the forward-looking statements. Revelation cautions readers not to place undue reliance on any such forward looking statements, which speak only as of the date they were made. The following factors, among others, could cause actual results to differ materially from those described in these forward-looking statements: the ability of Revelation to meet its financial and strategic goals, due to, among other things, competition; the ability of Revelation to grow and manage growth profitability and retain its key employees; the possibility that the Revelation may be adversely affected by other economic, business, and/or competitive factors; risks relating to the successful development of Revelation's product candidates; the ability to successfully complete planned clinical studies of its product candidates; the risk that we may not fully enroll our clinical studies or enrollment will take longer than expected; risks relating to the occurrence of adverse safety events and/or unexpected concerns that may arise from data or analysis from our clinical studies; changes in applicable laws or regulations; expected initiation of the clinical studies, the timing of clinical data; the outcome of the clinical data, including whether the results of such study is positive or whether it can be replicated; the outcome of data collected, including whether the results of such data and/or correlation can be replicated; the timing, costs, conduct and outcome of our other clinical studies; the anticipated treatment of future clinical data by the FDA, the EMA or other regulatory authorities, including whether such data will be sufficient for approval; the success of future development activities for its product candidates; potential indications for which product candidates may be developed; the ability of Revelation to maintain the listing of its securities on NASDAQ; the expected duration over which Revelation's balances will fund its operations; and other risks and uncertainties described herein, as well as those risks and uncertainties discussed from time to time in other reports and other public filings with the SEC by Revelation.
REVELATION BIOSCIENCES, INC.
Consolidated Statements of Operations
Three Months Ended | Nine Months Ended | |||||||||||
2025 | 2024 | 2025 | 2024 | |||||||||
Operating expenses: | ||||||||||||
Research and development | $ | 922,857 | $ | 830,981 | $ | 3,099,667 | $ | 2,943,492 | ||||
General and administrative | 1,020,154 | 965,705 | 3,399,559 | 3,277,729 | ||||||||
Total operating expenses | 1,943,011 | 1,796,686 | 6,499,226 | 6,221,221 | ||||||||
Loss from operations | (1,943,011 | ) | (1,796,686 | ) | (6,499,226 | ) | (6,221,221 | ) | ||||
Other income (expense): | ||||||||||||
Change in fair value of warrant liability | 611 | 6,041 | 2,071 | 78,884 | ||||||||
Other income (expense), net | 35,224 | (450,920 | ) | 94,512 | (7,170,480 | ) | ||||||
Total other income (expense), net | 35,835 | (444,879 | ) | 96,583 | (7,091,596 | ) | ||||||
Net loss | $ | (1,907,176 | ) | $ | (2,241,565 | ) | $ | (6,402,643 | ) | $ | (13,312,817 | ) |
Deemed dividends | (2,769,742 | ) | - | (5,951,528 | ) | - | ||||||
Net loss attributable to common stockholders | $ | (4,676,918 | ) | $ | (2,241,565 | ) | $ | (12,354,171 | ) | $ | (13,312,817 | ) |
Net loss per share, basic and diluted | $ | (1.77 | ) | $ | (40.15 | ) | $ | (9.76 | ) | $ | (354.05 | ) |
Weighted-average shares used to compute net loss per share, basic and diluted | 2,644,733 | 55,832 | 1,265,571 | 37,602 | ||||||||
REVELATION BIOSCIENCES, INC.
Consolidated Balance Sheets
September 30, | December 31, | |||
ASSETS | ||||
Current assets: | ||||
Cash and cash equivalents | $ | 12,708,489 | $ | 6,499,018 |
Prepaid expenses and other current assets | 123,934 | 66,699 | ||
Total current assets | 12,832,423 | 6,565,717 | ||
Property and equipment, net | 23,919 | 56,332 | ||
Total assets | $ | 12,856,342 | $ | 6,622,049 |
LIABILITIES AND STOCKHOLDERS' EQUITY | ||||
Current liabilities: | ||||
Accounts payable | $ | 1,034,951 | $ | 783,621 |
Accrued expenses | 819,381 | 1,127,800 | ||
Warrant liability | 175 | 2,246 | ||
Total current liabilities | 1,854,507 | 1,913,667 | ||
Total liabilities | 1,854,507 | 1,913,667 | ||
Commitments and Contingencies (Note 4) | ||||
Stockholders' equity: | ||||
Common Stock, $0.001 par value; 500,000,000 shares authorized at September 30, 2025 and December 31, 2024 and 3,585,972 and 174,104 issued and outstanding at September 30, 2025 and December 31, 2024, respectively | 3,586 | 174 | ||
Additional paid-in-capital | 57,906,530 | 45,213,846 | ||
Accumulated deficit | (46,908,281 | ) | (40,505,638 | ) |
Total stockholders' equity | 11,001,835 | 4,708,382 | ||
Total liabilities and stockholders' equity | $ | 12,856,342 | $ | 6,622,049 |
Company Contacts
Mike Porter
Investor Relations
Porter LeVay & Rose Inc.
Email: [email protected]
Chester Zygmont, III
Chief Financial Officer
Revelation Biosciences Inc.
Email: [email protected]
SOURCE: Revelation Biosciences, Inc.
View the original press release on ACCESS Newswire
Y.Aukaiv--AMWN
