
-
 Teenage wonder Sooryavanshi says criticism 'affects me a bit'
Teenage wonder Sooryavanshi says criticism 'affects me a bit'
-
Japan startup seeks approval of cat kidney disease treatment

-
 Technician dies installing stage for Shakira concert in Rio
Technician dies installing stage for Shakira concert in Rio
-
Cut off from the West, Muscovites rediscover Russian 'roots'

-
 'Joint venture in reverse': foreign carmakers seek edge with China partners
'Joint venture in reverse': foreign carmakers seek edge with China partners
-
Nations backing fossil fuel exit 'a new power': conference host Colombia

-
 Rockets thrash Lakers, Wembanyama triumphant on Spurs return
Rockets thrash Lakers, Wembanyama triumphant on Spurs return
-
ECB set to hold rates steady with eye on Iran crisis

-
 Team-first Kane propelling Bayern to glory as PSG showdown looms
Team-first Kane propelling Bayern to glory as PSG showdown looms
-
Pogacar vows to keep going until Seixas 'destroys' him

-
 From Adele to Raye, the UK school nurturing future stars
From Adele to Raye, the UK school nurturing future stars
-
Final talks begin on missing piece for pandemic treaty

-
 Oil rises, stocks swing as peace talk hopes wobble
Oil rises, stocks swing as peace talk hopes wobble
-
'Heartbroken' Xavi Simons out of World Cup and Spurs relegation fight

-
 North Korea's Kim reaffirms support for Russia's 'sacred' Ukraine war
North Korea's Kim reaffirms support for Russia's 'sacred' Ukraine war
-
Spurs win in Wembanyama return to take 3-1 lead over Trail Blazers

-
 As some hijabs come off in Iran, restrictions still in place
As some hijabs come off in Iran, restrictions still in place
-
Orangutan uses Indonesia canopy bridge in 'world first': NGO

-
 Dealing with the dead in the ruins of Sudan's war
Dealing with the dead in the ruins of Sudan's war
-
North Korea strengthens nuclear push as US flails in Middle East

-
 Stage set for Elon Musk's court battle with OpenAI
Stage set for Elon Musk's court battle with OpenAI
-
Caught between wars, US Afghan allies trapped in Qatar without safe exit

-
 British royals begin four-day US visit despite shooting
British royals begin four-day US visit despite shooting
-
Suspect in shooting at Trump press dinner to appear in court
-
 Macau Pass Unveils AI Payment Assistant to Upgrade Local Payment Capabilities
Macau Pass Unveils AI Payment Assistant to Upgrade Local Payment Capabilities
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 27

-
 Fitzpatrick brothers capture PGA Tour's Zurich Classic pairs crown
Fitzpatrick brothers capture PGA Tour's Zurich Classic pairs crown
-
Spurs win in Wembanyama return to take 3-1 lead on Trail Blazers

-
 Toulouse fall to first home defeat for a year
Toulouse fall to first home defeat for a year
-
Global military spending surges on insecurity: report

-
 Marseille see Champions League chance slip further away
Marseille see Champions League chance slip further away
-
Nelly Korda wins LPGA Chevron Championship

-
 Syrian court begins proceedings against Assad and allies
Syrian court begins proceedings against Assad and allies
-
Colombia road bombing death toll rises to 20

-
 Raptors top Cavs to pull level in NBA playoff series
Raptors top Cavs to pull level in NBA playoff series
-
Iran minister heads to Russia as talks remain stalled

-
 Rinku stars as Kolkata edge Lucknow in Super Over
Rinku stars as Kolkata edge Lucknow in Super Over
-
T'Wolves Edwards to miss several weeks - report

-
 Michael Jackson biopic debuts atop N. America box office
Michael Jackson biopic debuts atop N. America box office
-
King Charles state visit to US to go on as planned after shooting

-
 Inter pegged back by Torino as Serie A title charge hits bump in road
Inter pegged back by Torino as Serie A title charge hits bump in road
-
Mali junta in crisis after minister killed, key city 'captured'

-
 Dortmund down Freiburg to seal Champions League spot
Dortmund down Freiburg to seal Champions League spot
-
McFarlane hails Chelsea 'character' after FA Cup semi-final win

-
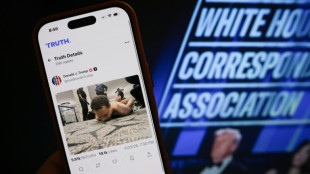
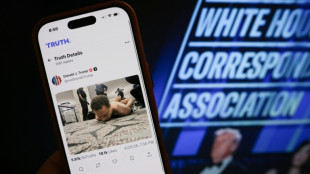 Gunman sought to kill Trump, cabinet at gala dinner
Gunman sought to kill Trump, cabinet at gala dinner
-
Arsenal punish Lyon errors in Champions League semi

-
 Suspect in US press gala shooting - what we know
Suspect in US press gala shooting - what we know
-
Key US senator lifts block on Fed chair nominee

-
 Attacks in Mali: What we know
Attacks in Mali: What we know
-
Vollering wins women's Lige-Bastogne-Liege for 3rd time


Moderna Receives European Commission Marketing Authorization for COVID-19 Vaccine mNEXSPIKE
mNEXSPIKE is the third Moderna vaccine authorized in the European Union, strengthening the Company's respiratory vaccine portfolio in Europe
CAMBRIDGE, MA / ACCESS Newswire / February 17, 2026 / Moderna, Inc. (NASDAQ:MRNA) today announced that the European Commission (EC) has granted marketing authorization for mNEXSPIKE® (mRNA-1283), a new COVID vaccine, for active immunization to prevent COVID-19 caused by SARS-CoV-2 in individuals aged 12 years and older.
This marketing authorization follows the positive opinion adopted by the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) and marks Moderna's third product authorized in the European Union, alongside Spikevax® and mRESVIA®, further strengthening the Company's respiratory vaccine portfolio in Europe. The marketing authorization is valid in all 27 European Union Member States, as well as Iceland, Liechtenstein and Norway. Moderna expects to make mNEXSPIKE available in Europe pending regulatory timelines and local market access pathways.
"We welcome the European Commission's decision, which reflects the strength of the scientific data supporting mNEXSPIKE and our continued commitment to advancing innovative vaccines for populations most at risk," said Stéphane Bancel, Chief Executive Officer of Moderna. "COVID-19 has become an endemic respiratory disease, with older adults continuing to bear a disproportionate burden of severe outcomes. Europe represents a key region for respiratory vaccines, and we are pleased to have a new vaccine available to help protect Europeans when the EU COVID market reopens starting this year in some markets."
The EC decision is supported by results from a randomized, observer-blind, active-controlled Phase 3 clinical trial (EudraCT: 2023-000884-30; ClinicalTrials.gov: NCT05815498), which enrolled approximately 11,400 participants aged 12 years and older. The primary efficacy objective of the study was to demonstrate non-inferior vaccine efficacy against COVID-19 starting 14 days after mNEXSPIKE compared to that after the comparator vaccine, mRNA-1273 (Spikevax), Moderna's original COVID-19 vaccine. In the trial, participants received either a 10 μg dose of mNEXSPIKE or a 50 μg dose of Spikevax. mNEXSPIKE showed a 9.3% higher relative vaccine efficacy compared to Spikevax in individuals aged 12 years and older, and in a descriptive subgroup analysis, a 13.5% higher relative vaccine efficacy in adults aged 65 years and older.
In the Phase 3 trial, mNEXSPIKE was found to have a similar safety profile to Spikevax, with fewer local reactions and comparable systemic reactions. The most commonly solicited adverse reactions were injection pain, fatigue, headache and myalgia.
Moderna has already received regulatory approval for mNEXSPIKE in the U.S., Canada and Australia, and continues to pursue approvals in additional markets worldwide.
About Moderna
Moderna is a pioneer and leader in the field of mRNA medicine. Through the advancement of its technology platform, Moderna is reimagining how medicines are made to transform how we treat and prevent diseases. Since its founding, Moderna's mRNA platform has enabled the development of vaccines and therapeutics across infectious diseases, cancer, rare diseases and more.
With a global team and a unique culture, driven by the company's values and mindsets, Moderna's mission is to deliver the greatest possible impact to people through mRNA medicines. For more information about Moderna, please visit modernatx.com and connect with us on X, Facebook, Instagram, YouTube and LinkedIn.
mNEXSPIKE®, Spikevax® and mRESVIA® are registered trademarks of Moderna.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including statements regarding: the availability of mNEXSPIKE in Europe; the opportunity in the European respiratory vaccine market and Moderna's geographical expansion; the safety profile of mNEXSPIKE; and potential approvals in additional markets worldwide. In some cases, forward-looking statements can be identified by terminology such as "will," "may," "should," "could," "expects," "intends," "plans," "aims," "anticipates," "believes," "estimates," "predicts," "potential," "continue," or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this press release are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond Moderna's control and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks, uncertainties, and other factors include, among others, those risks and uncertainties described under the heading "Risk Factors" in Moderna's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, filed with the U.S. Securities and Exchange Commission (SEC), and in subsequent filings made by Moderna with the SEC, which are available on the SEC's website at www.sec.gov. Except as required by law, Moderna disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release in the event of new information, future developments or otherwise. These forward-looking statements are based on Moderna's current expectations and speak only as of the date of this press release.
Moderna Contacts
Media:
Chris Ridley
Head of Global Media Relations
+1 617-800-3651
[email protected]
Investors:
Lavina Talukdar
Senior Vice President & Head of Investor Relations
+1 617-209-5834
[email protected]
SOURCE: Moderna, Inc.
View the original press release on ACCESS Newswire
D.Cunningha--AMWN