
-
 No Iran delegation sent to US talks yet as truce expiry nears
No Iran delegation sent to US talks yet as truce expiry nears
-
Rover discovers more building blocks of life on Mars

-
 Russia, North Korea connect road bridge ahead of summer opening
Russia, North Korea connect road bridge ahead of summer opening
-
'Strangled': Pakistan faces economic imperative in Iran war peace push

-
 Apple's Tim Cook to step down as CEO after 15-year run
Apple's Tim Cook to step down as CEO after 15-year run
-
Michael Jackson fans pack Hollywood for biopic premiere

-
 Turkey arrests 110 coal miners on hunger strike
Turkey arrests 110 coal miners on hunger strike
-
Oil prices dip, stocks rise on lingering Iran peace hopes

-
 Associated British Foods to spin off Primark clothes brand
Associated British Foods to spin off Primark clothes brand
-
Pope visits Eq. Guinea on last stop of Africa tour

-
 Hello Kitty's parent company to make own video games
Hello Kitty's parent company to make own video games
-
Di Matteo says 'vital' for faltering Chelsea to add experience

-
 Ex-Spurs star Davids condemns 'lack of quality, lack of management'
Ex-Spurs star Davids condemns 'lack of quality, lack of management'
-
Turkmenistan, the gas giant increasingly dependent on China

-
 Romanian AI music sensation Lolita sparks racism debate
Romanian AI music sensation Lolita sparks racism debate
-
Timberwolves battle back to stun Nuggets in NBA playoffs

-
 Eta appointment 'no surprise' for Union Berlin's ascendant women
Eta appointment 'no surprise' for Union Berlin's ascendant women
-
Democrats eye Virginia gains in war with Trump over US voting map

-
 Tourists trickle back to Kashmir, one year after deadly attack
Tourists trickle back to Kashmir, one year after deadly attack
-
Inside the world of ultra-luxury wedding cakes

-
 Chinese AI circuit board maker soars on Hong Kong debut
Chinese AI circuit board maker soars on Hong Kong debut
-
Oil prices dip, most stocks rise on lingering Iran peace hopes

-
 Tim Cook's time as Apple chief marked by profit absent awe
Tim Cook's time as Apple chief marked by profit absent awe
-
Mitchell, Harden shine as Cavs down Raptors for 2-0 series lead

-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Lobe Sciences Ltd. Engages Massive Intelligence Pty Ltd for Investor Relations and Corporate Communications Services

-
 Genoil Inc. Offers 30-60 Day Upstream Solution To Combat Hormuz Crisis As Physical Oil Tops $250, Threatening $700 Barrel Based On A Global Derivative Collapse
Genoil Inc. Offers 30-60 Day Upstream Solution To Combat Hormuz Crisis As Physical Oil Tops $250, Threatening $700 Barrel Based On A Global Derivative Collapse
-
Silver Range Advances the Drum Project in Utah

-
 Battery X Metals Files International PCT Patent Application for Lithium-Ion Battery Rebalancing Technology, Providing a Pathway to Pursue Patent Protection in 150+ Countries for Technology Validated by a Leading Scientific Institution to Recover ~99% Capacity Loss and Extend Battery Lifespan
Battery X Metals Files International PCT Patent Application for Lithium-Ion Battery Rebalancing Technology, Providing a Pathway to Pursue Patent Protection in 150+ Countries for Technology Validated by a Leading Scientific Institution to Recover ~99% Capacity Loss and Extend Battery Lifespan
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 21

-
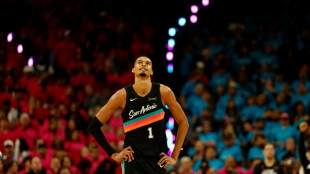 Wembanyama wins NBA defensive player of the year
Wembanyama wins NBA defensive player of the year
-
'The Devil Wears Prada 2' stars reunite for glamorous premiere

-
 El Salvador holds mass trial of nearly 500 alleged gang members
El Salvador holds mass trial of nearly 500 alleged gang members
-
Apple's Tim Cook to step down as CEO in September

-
 West Ham's draw at Palace relegates Wolves, piles pressure on Spurs
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs
-
Canadian tourist killed in Mexico archaeological site shooting

-
 Wolves relegated from Premier League
Wolves relegated from Premier League
-
Oil jumps on Hormuz tensions, stocks mostly retreat

-
 Colombian environmental activist honored amid threats and exile
Colombian environmental activist honored amid threats and exile
-
Gun battle traps more than 200 tourists at Rio viewpoint

-
 Alcaraz may skip French Open rather than rush injury comeback
Alcaraz may skip French Open rather than rush injury comeback
-
Top US court to hear case of Catholic schools excluded from state funding

-
 Trump Fed chair pick to vow interest rate independence at key hearing
Trump Fed chair pick to vow interest rate independence at key hearing
-
EU to host Taliban officials for talks on deporting Afghans

-
 Blue Origin probing rocket's failure to deliver satellite
Blue Origin probing rocket's failure to deliver satellite
-
Pope blasts 'exploitation' as he wraps up tour of Angola


IGC Pharma Highlights Alzheimer's Pipeline with Encouraging Preclinical Data on IGC-M3 and Advancing Phase 2 Cannabinoid-Based IGC-AD1
Positions IGC at the intersection of biotech innovation and cannabinoid science in one of the largest unmet medical needs in the world
Alzheimer's affects over 50 million people globally, with more than 400 million at risk from underlying disease pathology
POTOMAC, MARYLAND / ACCESS Newswire / August 12, 2025 / IGC Pharma, Inc. ("IGC" or the "Company") (NYSE American:IGC) today announced promising preclinical results for IGC-M3, a novel small molecule designed to address multiple biological drivers of Alzheimer's disease. IGC-M3 demonstrated activity in laboratory models against beta-amyloid aggregation, oxidative stress, mitochondrial dysfunction, and neuroinflammation, four key contributors to Alzheimer's pathology.
The findings underscore IGC Pharma's dual-pronged strategy in Alzheimer's disease, which includes:
IGC-AD1 - A proprietary, cannabinoid-based formulation currently in Phase 2 clinical trials for agitation in Alzheimer's disease, with potential disease-modifying effects under investigation.
IGC-M3 - A multifunctional, next-generation small molecule with the potential to impact multiple disease mechanisms, currently in preclinical development.
"Few companies in the Alzheimer's space are advancing both a late-stage cannabinoid-based therapy and an early-stage small molecule with multi-pathway potential," said Ram Mukunda, CEO of IGC Pharma. "IGC-AD1 offers a near-term opportunity to address agitation, a debilitating symptom affecting up to 76% of Alzheimer's patients using a combination of two actives one of which is low doses of Δ9-tetrahydrocannabinol ("THC"), a naturally extracted cannabinoid, while IGC-M3 expands our reach further into disease modification. Together, they position IGC as a differentiated player in one of the largest unmet medical needs in the world" With natural-cannabinoid based IGC-AD1 approaching critical Phase 2 milestones and synthetic IGC-M3 progressing through preclinical development, IGC Pharma is positioned to deliver multiple data-driven catalysts in FY2026 that could create lasting value for patients, caregivers, and shareholders alike.
Reducing Harmful Amyloid Clumps:
In biochemical assays, IGC-M3 prevented over 90% of the formation of beta-amyloid fibrils - a type of protein buildup linked to Alzheimer's - when tested at equal concentrations in the lab. It also helped break apart existing amyloid clumps in a dose-dependent way, with an effective concentration of about 7.9 micromolar. This suggests IGC-M3 may help keep these proteins in a safer, non-clumped form.
Balancing Metal Ions:
Parts of IGC-M3's structure allow it to bind certain metal ions, like copper and zinc, which have been linked to Alzheimer's disease. Lab tests confirmed that IGC-M3 can attach to these metals, which may help reduce metal-related oxidative stress and protein clumping in the brain.
Powerful Antioxidant Properties:
In antioxidant tests, IGC-M3 outperformed vitamin C in neutralizing harmful free radicals. It also helped protect human nerve cells from oxidative stress caused by hydrogen peroxide, improving their survival in the lab.
Protecting Mitochondria and Preventing Cell Death:
IGC-M3 reduced signs of mitochondrial damage caused by beta-amyloid, such as loss of membrane potential, increased oxidative molecules, and release of cytochrome c. It also lowered cell death rates without harming healthy cells.
Reducing Brain Inflammation:
In a model of brain immune cell activity, IGC-M3 lowered levels of a protein marker of inflammation, restored normal cell shape, and reduced stiffness and stickiness of the cells. It also decreased levels of inflammation-related molecules, including TNF-α, IL-6, and NF-κB.
Next Steps:
Although these findings are in vitro models, the breadth of biological effects observed suggests that M3 has the potential to modulate multiple pathways in the Alzheimer's disease cascade. The Company expects that M3 will advance to in vivo studies to evaluate potential efficacy and safety in animal models. If benefits are confirmed, M3 could represent a promising candidate for the development of disease-modifying treatments aimed at slowing or halting neurodegenerative progression in Alzheimer's disease.
About IGC Pharma (dba IGC):
IGC Pharma (NYSE American: IGC) is a clinical-stage biotechnology company leveraging AI to develop innovative treatments for Alzheimer's and metabolic disorders. Our lead asset, IGC-AD1, is a cannabinoid-based therapy currently in a Phase 2 trial (CALMA) for agitation in Alzheimer's dementia. Our pipeline includes TGR-63, targeting amyloid plaques, and early-stage programs focused on neurodegeneration, tau proteins, and metabolic dysfunctions. We integrate AI to accelerate drug discovery, optimize clinical trials, and enhance patient targeting. With 30 patent filings and a commitment to innovation, IGC Pharma is advancing breakthrough therapies.
Forward-Looking Statements:
This press release contains forward-looking statements. These forward-looking statements are based largely on IGC Pharma's expectations and are subject to several risks and uncertainties, certain of which are beyond IGC Pharma's control. Actual results could differ materially from these forward-looking statements as a result of, among other factors, the Company's failure or inability to commercialize one or more of the Company's products or technologies, including the products or formulations described in this release, or failure to obtain regulatory approval for the products or formulations, where required, or government regulations affecting AI or the AI algorithms not working as intended or producing accurate predictions; general economic conditions that are less favorable than expected; the FDA's general position regarding cannabis- and hemp-based products; and other factors, many of which are discussed in IGC Pharma's U.S. Securities and Exchange Commission ("SEC") filings. IGC incorporates by reference its Annual Report on Form 10-K filed with the SEC on June 27, 2025, as if fully incorporated and restated herein. Considering these risks and uncertainties, there can be no assurance that the forward-looking information contained in this release will occur. IGC Pharma, Inc. assumes no obligation to update forward-looking statements contained in this release as the result of new information or future events or developments.
Contact Information
Rosalyn Christian
IMS Investor Relations
[email protected]
(203) 972-9200
SOURCE: IGC Pharma, Inc.
View the original press release on ACCESS Newswire
L.Mason--AMWN