
-
 No Iran delegation sent to US talks yet as truce expiry nears
No Iran delegation sent to US talks yet as truce expiry nears
-
Rover discovers more building blocks of life on Mars

-
 Russia, North Korea connect road bridge ahead of summer opening
Russia, North Korea connect road bridge ahead of summer opening
-
'Strangled': Pakistan faces economic imperative in Iran war peace push

-
 Apple's Tim Cook to step down as CEO after 15-year run
Apple's Tim Cook to step down as CEO after 15-year run
-
Michael Jackson fans pack Hollywood for biopic premiere

-
 Turkey arrests 110 coal miners on hunger strike
Turkey arrests 110 coal miners on hunger strike
-
Oil prices dip, stocks rise on lingering Iran peace hopes

-
 Associated British Foods to spin off Primark clothes brand
Associated British Foods to spin off Primark clothes brand
-
Pope visits Eq. Guinea on last stop of Africa tour

-
 Hello Kitty's parent company to make own video games
Hello Kitty's parent company to make own video games
-
Di Matteo says 'vital' for faltering Chelsea to add experience

-
 Ex-Spurs star Davids condemns 'lack of quality, lack of management'
Ex-Spurs star Davids condemns 'lack of quality, lack of management'
-
Turkmenistan, the gas giant increasingly dependent on China

-
 Romanian AI music sensation Lolita sparks racism debate
Romanian AI music sensation Lolita sparks racism debate
-
Timberwolves battle back to stun Nuggets in NBA playoffs

-
 Eta appointment 'no surprise' for Union Berlin's ascendant women
Eta appointment 'no surprise' for Union Berlin's ascendant women
-
Democrats eye Virginia gains in war with Trump over US voting map

-
 Tourists trickle back to Kashmir, one year after deadly attack
Tourists trickle back to Kashmir, one year after deadly attack
-
Inside the world of ultra-luxury wedding cakes

-
 Chinese AI circuit board maker soars on Hong Kong debut
Chinese AI circuit board maker soars on Hong Kong debut
-
Oil prices dip, most stocks rise on lingering Iran peace hopes

-
 Tim Cook's time as Apple chief marked by profit absent awe
Tim Cook's time as Apple chief marked by profit absent awe
-
Mitchell, Harden shine as Cavs down Raptors for 2-0 series lead

-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Lobe Sciences Ltd. Engages Massive Intelligence Pty Ltd for Investor Relations and Corporate Communications Services

-
 Genoil Inc. Offers 30-60 Day Upstream Solution To Combat Hormuz Crisis As Physical Oil Tops $250, Threatening $700 Barrel Based On A Global Derivative Collapse
Genoil Inc. Offers 30-60 Day Upstream Solution To Combat Hormuz Crisis As Physical Oil Tops $250, Threatening $700 Barrel Based On A Global Derivative Collapse
-
Silver Range Advances the Drum Project in Utah

-
 Battery X Metals Files International PCT Patent Application for Lithium-Ion Battery Rebalancing Technology, Providing a Pathway to Pursue Patent Protection in 150+ Countries for Technology Validated by a Leading Scientific Institution to Recover ~99% Capacity Loss and Extend Battery Lifespan
Battery X Metals Files International PCT Patent Application for Lithium-Ion Battery Rebalancing Technology, Providing a Pathway to Pursue Patent Protection in 150+ Countries for Technology Validated by a Leading Scientific Institution to Recover ~99% Capacity Loss and Extend Battery Lifespan
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 21

-
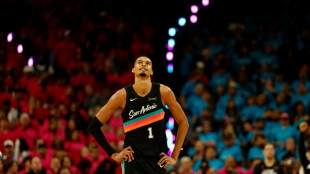 Wembanyama wins NBA defensive player of the year
Wembanyama wins NBA defensive player of the year
-
'The Devil Wears Prada 2' stars reunite for glamorous premiere

-
 El Salvador holds mass trial of nearly 500 alleged gang members
El Salvador holds mass trial of nearly 500 alleged gang members
-
Apple's Tim Cook to step down as CEO in September

-
 West Ham's draw at Palace relegates Wolves, piles pressure on Spurs
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs
-
Canadian tourist killed in Mexico archaeological site shooting

-
 Wolves relegated from Premier League
Wolves relegated from Premier League
-
Oil jumps on Hormuz tensions, stocks mostly retreat

-
 Colombian environmental activist honored amid threats and exile
Colombian environmental activist honored amid threats and exile
-
Gun battle traps more than 200 tourists at Rio viewpoint

-
 Alcaraz may skip French Open rather than rush injury comeback
Alcaraz may skip French Open rather than rush injury comeback
-
Top US court to hear case of Catholic schools excluded from state funding

-
 Trump Fed chair pick to vow interest rate independence at key hearing
Trump Fed chair pick to vow interest rate independence at key hearing
-
EU to host Taliban officials for talks on deporting Afghans

-
 Blue Origin probing rocket's failure to deliver satellite
Blue Origin probing rocket's failure to deliver satellite
-
Pope blasts 'exploitation' as he wraps up tour of Angola


Tharimmune Secures Key Global Patents for TH-104, Laying Foundation for Broader Indications Following Initial Development as a National Security Medical Countermeasure Against Weaponized Fentanyl
RED BANK, NEW JERSEY / ACCESS Newswire / August 13, 2025 / Tharimmune, Inc. (Nasdaq:THAR) ("Tharimmune" or the "Company"), a clinical-stage biotechnology company dedicated to developing innovative therapeutic candidates for inflammation, immunology, and critical unmet medical needs, today announced a significant expansion of its intellectual property portfolio for TH-104, its novel transmucosal film. The new patents, recently granted and allowed across key global markets, will enable the Company's long-term strategy of pursuing broader indications for the technology, beginning with its lead program as a critical medical countermeasure for military and first responders against high-potency opioids.
This announcement follows recent positive feedback from the U.S. Food and Drug Administration (FDA) regarding the New Drug Application (NDA) pathway for TH-104. The FDA confirmed that a 505(b)(2) NDA submission for the prevention of respiratory and/or central nervous system distress is a viable path, and no additional clinical trials seem to be required for this specific indication for use in military personnel and/or first responders who may be exposed to weaponized high-potency opioids, such as fentanyl or its analogues. This positive feedback, combined with the appointment of counter-terrorism expert James Liddy to the Board of Directors, accelerates the Company's efforts to position TH-104 as a national security solution.
The recent granting and allowance of patents in Japan, Mexico, Australia, and the United States further strengthen the Company's intellectual property position, providing protection for TH-104 out to at least 2040 in key markets. While the initial focus is on military and first responders, these patents are foundational for addressing other significant medical needs to combat pruritus, a debilitating form of itch associated with certain chronic liver and kidney diseases.
TH-104 is a buccal film formulation of nalmefene, an opioid antagonist with a significantly longer half-life compared to existing options of autoinjectors and other formulations containing the active ingredient of products such as Narcan®. This extended duration may provide sustained protection, which is crucial for personnel operating in high-risk environments where repeated dosing may not be feasible.
"The recent patent grants are important for our TH-104 platform," said Sireesh Appajosyula, CEO of Tharimmune. "This is a dual achievement which validates our innovative drug delivery technology and secures the long-term potential of TH-104 to address multiple high-impact indications. While we are prioritizing its role as a life-saving medical countermeasure for our military and first responders, this patent portfolio provides a runway to develop our lead candidate for other debilitating conditions where there is a significant unmet medical need."
The newly granted and allowed patents specifically cover a novel method and device for transmucosal administration of nalmefene via a single-layer, self-supporting, mucoadhesive film with distinct domains for optimized drug delivery and pH balance.
The granted and allowed patents include:
United States (US) Application No. 17/285,971 (Allowed)
Japan (JP) Patent No. 7603583 (Granted)
Mexico (MX) Patent No. 422769 (Granted)
Australia (AU) Patent Application No. 2019361980 (Allowed)
Tharimmune is dedicated to advancing TH-104 to provide a potentially effective and patient-friendly treatment as an option for those in need, beginning with its critical role in national security.
About Tharimmune, Inc.
Tharimmune is a clinical-stage biotechnology company developing a diverse portfolio of therapeutic candidates in immunology, inflammation and oncology. Its lead clinical asset, TH104, is being developed for a specific indication via a 505(b)2 pathway for respiratory and/or nervous system depression in military personnel and chemical incident responders who may encounter environments contaminated with high-potency opioids. The expanded pipeline includes other indications for TH104, such as chronic pruritus in primary biliary cholangitis and TH023, a new approach to treating autoimmune diseases along with an early-stage multispecific biologic platform targeting unique epitopes against multiple solid tumors through its proprietary EpiClick™ Technology. The Company has a license agreement with OmniAb, Inc. to access their antibody discovery technology for targeting specified disease markers. Tharimmune continues to position itself as a leader in patient-centered innovation while working to deliver long-term value for shareholders. For more information, visit: www.tharimmune.com.
Forward Looking Statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, contained in this press release, including statements regarding the timing and design of Tharimmune's future Phase 2 trial, Tharimmune's strategy, future operations, future financial position, projected costs, prospects, plans and objectives of management, are forward-looking statements. The words "anticipate," "believe," "continue," "could," "depends," "estimate," "expect," "intend," "may," "ongoing," "plan," "potential," "predict," "project," "target," "should," "will," "would," and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. The Company may not actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements, and you should not place undue reliance on these forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in these forward-looking statements. Factors that may cause such differences, include, but are not limited to, those discussed under Risk Factors set forth in our Annual Report on Form 10-K for the year ended December 31, 2024 and other periodic reports filed by the Company from time to time with the Securities and Exchange Commission. In addition, the forward-looking statements included in this press release represent the Company's views as of the date of this release. Subsequent events and developments may cause the Company's views to change; however, the Company does not undertake and specifically disclaims any obligation to update or revise any forward-looking statements to reflect new information, future events or circumstances or to reflect the occurrences of unanticipated events, except as may be required by applicable law. These forward-looking statements should not be relied upon as representing the Company's views as of any date subsequent to the date of this release.
Contacts:
Tharimmune, Inc.
[email protected]
SOURCE: Tharimmune Inc.
View the original press release on ACCESS Newswire
M.A.Colin--AMWN