-
 Venezuela's Machado doesn't regret gifting Nobel Peace Prize to Trump
Venezuela's Machado doesn't regret gifting Nobel Peace Prize to Trump
-
No date set for next round of Iran-US talks: Iran deputy FM

-
 Iran closes Hormuz Strait again over US blockade, ships reverse course
Iran closes Hormuz Strait again over US blockade, ships reverse course
-
'We've already beaten other favorites', Lyon's Endrick warns PSG

-
 Turkey says Israel using security as a pretext to acquire 'more land'
Turkey says Israel using security as a pretext to acquire 'more land'
-
Iran closes Hormuz Strait again over US blockade with ships mid-transit

-
 French film star Nathalie Baye dead at 77: family to AFP
French film star Nathalie Baye dead at 77: family to AFP
-
China sex toy makers cautiously embrace AI wave

-
 Paramount's CinemaCon charm offensive gets lukewarm reception
Paramount's CinemaCon charm offensive gets lukewarm reception
-
Game over: Players press EU to ban 'destroying' video titles

-
 Churches to the rescue of Cuba's legions of poor
Churches to the rescue of Cuba's legions of poor
-
In Trump era, fearful left-leaning Americans turn to guns

-
 Pope brings Africa tour to Angola as Trump feud drags on
Pope brings Africa tour to Angola as Trump feud drags on
-
Fitzpatrick charges to one-shot lead at RBC Heritage

-
 Andreeva sinks Swiatek to meet top seed Rybakina in Stuttgart semis
Andreeva sinks Swiatek to meet top seed Rybakina in Stuttgart semis
-
Carrick won't rule out Rashford return to Man Utd

-
 Lampard restores reputation by leading Coventry to Premier League
Lampard restores reputation by leading Coventry to Premier League
-
'Gouged': World Cup fans to pay 'insane' $150 for NY stadium train ticket

-
 Lens leave it late to edge Toulouse and keep pressure on PSG
Lens leave it late to edge Toulouse and keep pressure on PSG
-
Inter swat aside Cagliari to continue Serie A title procession

-
 Thunder stay in the moment as NBA title repeat beckons
Thunder stay in the moment as NBA title repeat beckons
-
US Catholics unsettled by Trump's feud with pope

-
 US Supreme Court sides with Chevron in environmental case
US Supreme Court sides with Chevron in environmental case
-
World Cup fans to pay $150 for NY stadium train ticket: official

-
 Gujarat's Gill consigns Kolkata to fifth defeat in IPL
Gujarat's Gill consigns Kolkata to fifth defeat in IPL
-
Top takeaways from CinemaCon: the year's hottest movies

-
 Lebanon president says working on 'permanent agreements' after Israel truce
Lebanon president says working on 'permanent agreements' after Israel truce
-
Top-seeded Pistons embrace underdog tag

-
 Andreeva sinks Swiatek to reach Stuttgart semis
Andreeva sinks Swiatek to reach Stuttgart semis
-
Genital mutilation: the silent suffering of Colombia's Indigenous girls

-
 UEFA probe after photographers injured at Bayern-Real game
UEFA probe after photographers injured at Bayern-Real game
-
Trump tells AFP 'no sticking points' for deal with Iran

-
 Trump tells AFP Iran deal close, 'no sticking points' left
Trump tells AFP Iran deal close, 'no sticking points' left
-
Shippers eye Iran Hormuz reopening with wariness

-
 France, UK to lead 'defensive' force for Hormuz
France, UK to lead 'defensive' force for Hormuz
-
Fils takes out Musetti to reach Barcelona Open semis

-
 Griezmann soaking up last Atletico moments before 'joy' of Copa final
Griezmann soaking up last Atletico moments before 'joy' of Copa final
-
Polish stadium cancels Kanye West concert

-
 Lille's Bentaleb out after 'minor surgery' for infection
Lille's Bentaleb out after 'minor surgery' for infection
-
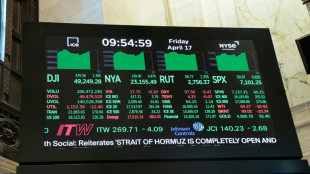
Oil plunges, stocks jump as Iran declares Hormuz open
-
 Trump signals Iran deal near, hails 'brilliant day for world'
Trump signals Iran deal near, hails 'brilliant day for world'
-
Zverev fights past Cerundolo to reach Munich semis

-
 France, UK to lead multinational Hormuz mission
France, UK to lead multinational Hormuz mission
-
Vondrousova in trouble after shutting door on doping officer

-
 Stranded seafarers endure costly path home from Gulf
Stranded seafarers endure costly path home from Gulf
-
Iran declares Hormuz open as Lebanon ceasefire begins

-
 Pope Leo comes into his own with Trump spat
Pope Leo comes into his own with Trump spat
-
Alcaraz withdraws from Madrid Masters after wrist injury

-
 Arteta tells spluttering Arsenal to embrace title pressure ahead of Man City showdown
Arteta tells spluttering Arsenal to embrace title pressure ahead of Man City showdown
-
Chelsea star Caicedo signs seven-year contract extension


FDA approves Omisirge(R) as First Approved Cell Therapy to Treat Severe Aplastic Anemia
Study demonstrated early and sustained hematopoietic recovery and transfusion independence
Omisirge is a new and novel transplant option for patients with SAA
Additional manufacturing capacity for Omisirge will be established in the U.S.
Represents a supplementary FDA approval for Omisirge
DUBLIN, IE AND LONDON, UK / ACCESS Newswire / December 5, 2025 / Ayrmid Ltd. ("Ayrmid" or the "Company"), the parent company of Gamida Cell Inc., a leader in cell therapy innovation announces that it has received U.S. Food and Drug Administration ("FDA") approval for Omisirge (Omidubicel-onlv) for the treatment of Severe Aplastic Anemia (SAA).
This approval is based on the ongoing open-label, single-center study, led by Dr. Richard Childs of the National Heart, Lung, and Blood Institute (NHLBI) at the National Institutes of Health (NIH), which demonstrated highly encouraging results including:
Median time to neutrophil recovery: 11 days
86% of patients had early and sustained neutrophil recovery at 100 days
Immune recovery post-transplant was rapid
86% of patients achieved red blood cell transfusion independence
No cases of BMT-CTN severe acute graft-versus-host disease (GVHD), or chronic GVHD observed
92% disease-free and overall survival rate
Dr. Richard Childs of the National Heart, Lung, and Blood Institute (NHLBI) at the National Institutes of Health (NIH) commented: "The approval of Omisirge is a significant step forward in the treatment options available for patients with a high unmet medical need. Patients in the study with aplastic anemia were high risk, but had significantly better than expected outcomes and demonstrated remarkably fast and high rates of neutrophil engraftment. This was achieved with low rates of mild acute GVHD and no chronic GVHD meaning the patients achieved a rapid return to a normal life."
Dr. Joe Wiley, Ayrmid Chairman and Chief Executive Officer commented: "Alongside its initial FDA approval in hematological malignancy in 2023, Omisirge is a new and novel transplant option for the many SAA patients who do not have a matched related donor. This approval is potentially transformative for patients living with SAA, given the rapid and sustained recovery of blood counts observed. At Ayrmid, we remain deeply committed to advancing innovative therapies to help patients with serious unmet medical needs."
To meet the expected demand from SAA patients, Ayrmid has a strategic partnership in place with RoslinCT for the production of Omisirge in the U.S. alongside existing manufacturing facilities. The technology transfer and commercial manufacturing of Omisirge will take place in RoslinCT's state-of-the-art, purpose-built cell therapy CGMP manufacturing facility in Hopkinton, MA. beginning in 2027.
About Severe Aplastic Anemia
Severe Aplastic Anemia is a rare, life-threatening hematologic disorder in which the bone marrow fails to produce sufficient blood cells. Stem cell transplantation offers a potential cure; however, many patients lack a matched sibling donor. Ayrmid is advancing therapies to address this unmet medical need.
About Ayrmid Ltd. and Gamida Cell
Ayrmid Ltd. is the parent company of Gamida Cell Inc., a pioneering cell therapy company developing novel treatments designed to turn cells into powerful therapeutics. Gamida Cell Inc. currently has two FDA approved products on the market in the US, namely Omisirge (please see the current full Prescribing Information, including boxed warning, here) and APHEXDA® (please see the current full Prescribing Information here). Gamida Cell operates as a wholly owned subsidiary of Ayrmid Limited, a UK entity. For additional information, please visit www.gamida-cell.com or follow Gamida Cell on LinkedIn, X, Facebook or Instagram.
Contacts: Media, Investors / Business Development: [email protected]
This information is provided by RNS, the news service of the London Stock Exchange. RNS is approved by the Financial Conduct Authority to act as a Primary Information Provider in the United Kingdom. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com
SOURCE: Ayrmid Pharma Ltd.
View the original press release on ACCESS Newswire
D.Moore--AMWN