
-
 Bullying claims 'nonsense', actress Rebel Wilson tells Sydney court
Bullying claims 'nonsense', actress Rebel Wilson tells Sydney court
-
BP reports huge profit rise in first quarter

-
 Crude extends gains, stocks drop as Trump considers latest Iran proposal
Crude extends gains, stocks drop as Trump considers latest Iran proposal
-
How China block of AI deal could stop 'Singapore-washing'

-
 North Korean executions rose dramatically during Covid: report
North Korean executions rose dramatically during Covid: report
-
Budget airlines first to cut flights as jet fuel prices soar

-
 Simeone, Atletico chasing redemption against Arsenal
Simeone, Atletico chasing redemption against Arsenal
-
'Bring it on', says Rice as Arsenal chase Champions League history

-
 US says examining latest Iran proposal
US says examining latest Iran proposal
-
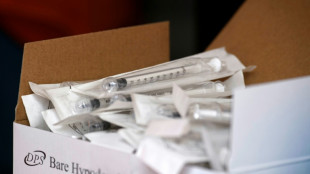
S. Korea probes syringe hoarding as war hits plastic makers
-
 Australia aims to tax tech giants unless they pay news outlets
Australia aims to tax tech giants unless they pay news outlets
-
Bangladesh's tigers stalk uncertain future in Sundarbans

-
 Horses unlikely saviours for those who serve in uniform
Horses unlikely saviours for those who serve in uniform
-
Crude extends gains as Trump considers latest Iran proposal

-
 Nations to kick off world-first fossil fuel exit talks
Nations to kick off world-first fossil fuel exit talks
-
Philippine museum brings deadly, lucrative galleon trade to life

-
 Opening remarks Tuesday in Elon Musk versus OpenAI
Opening remarks Tuesday in Elon Musk versus OpenAI
-
New York restaurant's $40 half chicken fuels cost of dining debate

-
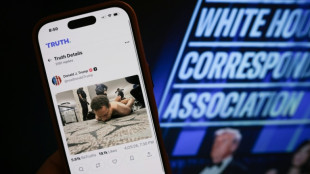 Trump shooting scare renews 'staged' conspiracy theory
Trump shooting scare renews 'staged' conspiracy theory
-
LIV Golf postpones June event set for New Orleans: reports

-
 Kingfisher Metals Confirms Early Jurassic Porphyry Mineralization Across Hank-Mary District, a Fertile Window Between Mitchell Cu-Au and Brucejack Au-Ag Deposits
Kingfisher Metals Confirms Early Jurassic Porphyry Mineralization Across Hank-Mary District, a Fertile Window Between Mitchell Cu-Au and Brucejack Au-Ag Deposits
-
Star Copper Completes Copperline Project Inaugural 2025 Field Program at Omineca Mining Division, BC

-
 BioNxt Applies Advanced Drug Delivery Strategy and Psychedelic Compound Library to Emerging Therapeutics Market Amid Accelerating Global Momentum
BioNxt Applies Advanced Drug Delivery Strategy and Psychedelic Compound Library to Emerging Therapeutics Market Amid Accelerating Global Momentum
-
SphingoTec Introduces ELISA sphingotest(R) penKid(R) to Enable Broad Kidney Biomarker Testing for Research Applications

-
 Apex Drills 4.02% REO over 23.7 m, within Broader 124 m Zone at 1.94% REO, Extending Mineralization Significantly to the South
Apex Drills 4.02% REO over 23.7 m, within Broader 124 m Zone at 1.94% REO, Extending Mineralization Significantly to the South
-
Hemogenyx Pharmaceuticals PLC Announces Institutional Fundraise

-
 Caledonia Mining Corporation Plc: Notice of Q1 2026 Results and Investor Presentation
Caledonia Mining Corporation Plc: Notice of Q1 2026 Results and Investor Presentation
-
Genflow Biosciences PLC Announces Expansion of Confidentiality Agreements

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 28
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 28
-
Colombian peace accord failed to protect nature: ex-leader Santos

-
 Nations have chance to break 'fossil fuel mindset': Mary Robinson
Nations have chance to break 'fossil fuel mindset': Mary Robinson
-
Colombia in mourning after deadliest attack in decades

-
 Jury in place for Elon Musk's legal battle with OpenAI
Jury in place for Elon Musk's legal battle with OpenAI
-
Weinstein rape accuser gives emotional testimony at US retrial

-
 Rybakina crashes out of Madrid Open, Sabalenka reaches quarters
Rybakina crashes out of Madrid Open, Sabalenka reaches quarters
-
Trump and team renew attacks on adversaries after gala shooting

-
 Carrick hails Casemiro and Fernandes after vital Man Utd win
Carrick hails Casemiro and Fernandes after vital Man Utd win
-
Felix, 40, says she plans comeback for LA Olympics

-
 French FM says Iran must make 'major concessions' to end crisis
French FM says Iran must make 'major concessions' to end crisis
-
Trains collide near Jakarta, killing five, injuring dozens

-
 Britain's King Charles meets Trump in bid to salvage ties
Britain's King Charles meets Trump in bid to salvage ties
-
Accused media gala gunman charged with attempting to assassinate Trump

-
 Man Utd beat Brentford to close on Champions League berth
Man Utd beat Brentford to close on Champions League berth
-
Third suspect pleads guilty in US murder of Jam Master Jay

-
 Milei bars media from presidential palace
Milei bars media from presidential palace
-
California billionaire tax appears headed to the ballot

-
 Trains collide near Jakarta, killing four, injuring dozens
Trains collide near Jakarta, killing four, injuring dozens
-
Kompany hails Kane, 'ageing like fine wine' as Bayern face PSG in Champions League

-
 UK's King Charles arrives in US to shore up Trump ties
UK's King Charles arrives in US to shore up Trump ties
-
Tuareg rebels in control of key Mali town


SphingoTec Introduces ELISA sphingotest(R) penKid(R) to Enable Broad Kidney Biomarker Testing for Research Applications
New ELISA sphingotest® penKid® enables widespread measurement of the kidney function biomarker Proenkephalin 119-159 (penKid) using standard laboratory equipment.
Only available test to match SphingoTec's reference chemiluminescence assay, developed with patented high-sensitivity technology to provide consistent results across platforms.
HENNIGSDORF, DE / ACCESS Newswire / April 28, 2026 / SphingoTec GmbH announces the launch of the ELISA sphingotest® penKid®, a new assay designed to make testing of its proprietary biomarker broadly accessible to research laboratories and pharmaceutical partners. Providing ease-of-use and precision, the test facilitates large-scale investigations of human kidney function in both acute and broader clinical research contexts.
Responding to growing research interest
The launch follows increasing demand from the scientific community to study penKid - a biomarker reflecting the current state of kidney function in critical care environments. While SphingoTec's high-sensitivity sphingotest® penKid® assay serves as the reference method for clinical research and third-party IVD assays, the assay technology requiring dedicated equipment was not easily adoptable in research laboratories.
The new ELISA sphingotest® penKid® now allows researchers worldwide to benefit from SphingoTec's patented detection technology using standard photometers, extending penKid testing capability to a wider range of laboratories.
Proven performance and data continuity
The ELISA sphingotest® penKid® has been developed to achieve excellent correlation with SphingoTec's reference chemiluminescence assay, ensuring consistent, high-quality results across different assay platforms. Both methods share the same underlying technology capable of detecting penKid concentrations in the picomolar range.
Thanks to this technology transfer, researchers can rely on the same analytical precision and reliability that supported the clinical studies establishing penKid as a valuable kidney function biomarker. The assay's robust design and German manufacturing ensure durable consistency and reproducibility.
With this research-use ELISA, SphingoTec responds to the increasing availability of non-validated assays and encourages researchers to not compromise assay quality and performance to ensure trustworthy results - since data from non-validated tests may reflect assay limitations rather than the true performance of the penKid biomarker.
Complementary approaches for clinical use and scientific exploration
SphingoTec pursues its commercial strategy for the biomarker penKid through strategic out-licensing partnerships such as Boditech Med, which has developed an IVDR-certified assay for routine clinical use and rapid diagnostics. The newly launched ELISA sphingotest® penKid® represents a complementary initiative, specifically designed to support high-throughput clinical and translational research. Through this dual approach, SphingoTec reaffirms its commitment to fostering scientific collaboration and promoting the broader exploration of penKid across diverse research settings.
"Developing the ELISA sphingotest® penKid® reflects our long-term commitment to improving critical care diagnostics," said Deborah Bergmann, Managing Director and CEO of SphingoTec GmbH. "Beyond its currently validated clinical applications, we see strong potential for penKid to support research and improve diagnostics in other fields where kidney function is relevant. By encouraging scientists worldwide to incorporate penKid into their studies and clinical programs, we aim to accelerate innovation and advance best-fit diagnostic solutions that ultimately improve patient care."
About SphingoTec
SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com
Media Contact:
Email: [email protected]
Phone +49-3302-20565-0
SphingoTec GmbH
Neuendorfstr. 15A
16761 Hennigsdorf, Germany
SOURCE: SphingoTec GmbH
View the original press release on ACCESS Newswire
Y.Nakamura--AMWN
