
-
 BP reports huge profit rise in first quarter
BP reports huge profit rise in first quarter
-
Crude extends gains, stocks drop as Trump considers latest Iran proposal

-
 How China block of AI deal could stop 'Singapore-washing'
How China block of AI deal could stop 'Singapore-washing'
-
North Korean executions rose dramatically during Covid: report

-
 Budget airlines first to cut flights as jet fuel prices soar
Budget airlines first to cut flights as jet fuel prices soar
-
Simeone, Atletico chasing redemption against Arsenal

-
 'Bring it on', says Rice as Arsenal chase Champions League history
'Bring it on', says Rice as Arsenal chase Champions League history
-
US says examining latest Iran proposal

-
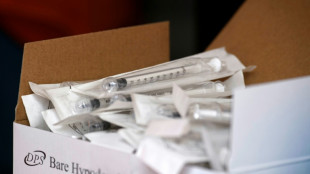 S. Korea probes syringe hoarding as war hits plastic makers
S. Korea probes syringe hoarding as war hits plastic makers
-
Australia aims to tax tech giants unless they pay news outlets

-
 Bangladesh's tigers stalk uncertain future in Sundarbans
Bangladesh's tigers stalk uncertain future in Sundarbans
-
Horses unlikely saviours for those who serve in uniform

-
 Crude extends gains as Trump considers latest Iran proposal
Crude extends gains as Trump considers latest Iran proposal
-
Nations to kick off world-first fossil fuel exit talks

-
 Philippine museum brings deadly, lucrative galleon trade to life
Philippine museum brings deadly, lucrative galleon trade to life
-
Opening remarks Tuesday in Elon Musk versus OpenAI

-
 New York restaurant's $40 half chicken fuels cost of dining debate
New York restaurant's $40 half chicken fuels cost of dining debate
-
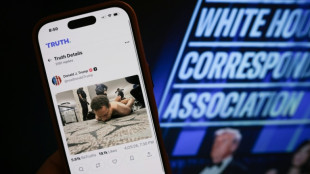
Trump shooting scare renews 'staged' conspiracy theory
-
 LIV Golf postpones June event set for New Orleans: reports
LIV Golf postpones June event set for New Orleans: reports
-
Star Copper Completes Copperline Project Inaugural 2025 Field Program at Omineca Mining Division, BC

-
 BioNxt Applies Advanced Drug Delivery Strategy and Psychedelic Compound Library to Emerging Therapeutics Market Amid Accelerating Global Momentum
BioNxt Applies Advanced Drug Delivery Strategy and Psychedelic Compound Library to Emerging Therapeutics Market Amid Accelerating Global Momentum
-
SphingoTec Introduces ELISA sphingotest(R) penKid(R) to Enable Broad Kidney Biomarker Testing for Research Applications

-
 Apex Drills 4.02% REO over 23.7 m, within Broader 124 m Zone at 1.94% REO, Extending Mineralization Significantly to the South
Apex Drills 4.02% REO over 23.7 m, within Broader 124 m Zone at 1.94% REO, Extending Mineralization Significantly to the South
-
Hemogenyx Pharmaceuticals PLC Announces Institutional Fundraise

-
 Caledonia Mining Corporation Plc: Notice of Q1 2026 Results and Investor Presentation
Caledonia Mining Corporation Plc: Notice of Q1 2026 Results and Investor Presentation
-
Genflow Biosciences PLC Announces Expansion of Confidentiality Agreements

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 28
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 28
-
Colombian peace accord failed to protect nature: ex-leader Santos

-
 Nations have chance to break 'fossil fuel mindset': Mary Robinson
Nations have chance to break 'fossil fuel mindset': Mary Robinson
-
Colombia in mourning after deadliest attack in decades

-
 Jury in place for Elon Musk's legal battle with OpenAI
Jury in place for Elon Musk's legal battle with OpenAI
-
Weinstein rape accuser gives emotional testimony at US retrial

-
 Rybakina crashes out of Madrid Open, Sabalenka reaches quarters
Rybakina crashes out of Madrid Open, Sabalenka reaches quarters
-
Trump and team renew attacks on adversaries after gala shooting

-
 Carrick hails Casemiro and Fernandes after vital Man Utd win
Carrick hails Casemiro and Fernandes after vital Man Utd win
-
Felix, 40, says she plans comeback for LA Olympics

-
 French FM says Iran must make 'major concessions' to end crisis
French FM says Iran must make 'major concessions' to end crisis
-
Trains collide near Jakarta, killing five, injuring dozens

-
 Britain's King Charles meets Trump in bid to salvage ties
Britain's King Charles meets Trump in bid to salvage ties
-
Accused media gala gunman charged with attempting to assassinate Trump

-
 Man Utd beat Brentford to close on Champions League berth
Man Utd beat Brentford to close on Champions League berth
-
Third suspect pleads guilty in US murder of Jam Master Jay

-
 Milei bars media from presidential palace
Milei bars media from presidential palace
-
California billionaire tax appears headed to the ballot

-
 Trains collide near Jakarta, killing four, injuring dozens
Trains collide near Jakarta, killing four, injuring dozens
-
Kompany hails Kane, 'ageing like fine wine' as Bayern face PSG in Champions League

-
 UK's King Charles arrives in US to shore up Trump ties
UK's King Charles arrives in US to shore up Trump ties
-
Tuareg rebels in control of key Mali town

-
 US Supreme Court hears Bayer bid to end Roundup weedkiller suits
US Supreme Court hears Bayer bid to end Roundup weedkiller suits
-
Separate goals, common enemy for Mali's jihadists and separatists


Hemogenyx Pharmaceuticals PLC Announces Institutional Fundraise
THIS ANNOUNCEMENT CONTAINS INSIDE INFORMATION AS STIPULATED UNDER THE UK VERSION OF THE MARKET ABUSE REGULATION NO 596/2014 WHICH IS PART OF ENGLISH LAW BY VIRTUE OF THE EUROPEAN (WITHDRAWAL) ACT 2018, AS AMENDED. ON PUBLICATION OF THIS ANNOUNCEMENT VIA A REGULATORY INFORMATION SERVICE, THIS INFORMATION IS CONSIDERED TO BE IN THE PUBLIC DOMAIN.
Hemogenyx Secures £3,000,000 to accelerate its Phase 1 Clinical Trials and initiate early commercialization of HG-CT-1
LONDON, UK / ACCESS Newswire / April 28, 2026 / Hemogenyx Pharmaceuticals plc (LSE:HEMO) is pleased to announce that it has raised £3,000,000 from a small group of institutional investors to support the acceleration of its clinical development programmes. The fundraising was completed through a direct subscription for 374,532 new ordinary shares in the Company at a market bid price at closing of trading on April 27 of £8.01 per share.
The net proceeds from the fundraising will be used primarily to accelerate the Phase I clinical trials of the Company's proprietary Chimeric Antigen Receptor T-cell therapy, HG-CT-1. This therapy is being developed for the treatment of relapsed/refractory acute myeloid leukaemia ("R/R AML") in adults and is now also being extended into paediatric patients.
Early Commercialisation Initiative
In September 2025, the Company announced that it had begun exploring the potential for early commercialisation of HG-CT-1 through the hospital exemption pathway under Estonia's Medicinal Products Act. The Company is pleased to report that the necessary hardware and infrastructure for the manufacturing of HG-CT-1 in Estonia have now been established.
Hemogenyx, in collaboration with its Estonian partner Cellin Technologies OÜ, intends to commence the technology transfer process required for local manufacturing of HG-CT-1 imminently.
The successful completion of this fundraising provides the Company with additional financial resources to advance its clinical programmes and progress its early commercialisation strategy for HG-CT-1. Further updates will be provided in due course.
An application is being made to the Main Market of the London Stock Exchange, and admission of the Placing Shares to trading is expected on or around May 6, 2026 ("Admission"). The Placing Shares will rank pari passu with the Company's existing Ordinary Shares.
Total Voting Rights
For the purpose of the Disclosure Guidance and Transparency Rules, following Admission the enlarged issued share capital of the Company will comprise 6,799,961 ordinary shares. The Company does not hold any shares in treasury. The above figure may be used by shareholders as the denominator for the calculations by which they will determine if they are required to notify their interest in, or a change to their interest in, the Company, under the Disclosure Guidance and Transparency Rules.
Dr Vladislav Sandler, CEO & Co-Founder of Hemogenyx Pharmaceuticals, commented:
"This fundraising marks an important step forward for Hemogenyx as we continue to advance HG-CT-1 through clinical development and move closer to potential early commercialisation. We are pleased to have secured the support of institutional investors who share our vision of bringing innovative, life-saving therapies to patients with relapsed or refractory acute myeloid leukaemia, including children.
The establishment of manufacturing infrastructure in Estonia and our planned technology transfer with Cellin Technologies represent key milestones in our strategy to accelerate patient access through the hospital exemption pathway. We believe this dual approach of progressing clinical trials while preparing for early commercialisation uniquely positions the Company to create value and address significant unmet medical needs."
UK Market Abuse Regulation (UK MAR) Disclosure
Certain information contained in this announcement would have been inside information for the purposes of Article 7 of Regulation No 596/2014 (as it forms part of UK domestic law by virtue of the European Union (Withdrawal) Act 2018) until the release of this announcement. The person responsible for arranging for the release of this announcement on behalf of Hemogenyx Pharmaceuticals plc is Dr Vladislav Sandler, Chief Executive Officer & Co-Founder.
Enquiries:
Hemogenyx Pharmaceuticals plc | |
Dr Vladislav Sandler, Chief Executive Officer & Co-Founder | |
Peter Redmond, Director | |
SP Angel Corporate Finance LLP | Tel: +44 (0)20 3470 0470 |
Matthew Johnson, Vadim Alexandre, Adam Cowl | |
AlbR Capital Limited | Tel: +44 (0)20 7469 0930 |
Lucy Williams, Duncan Vasey, Charles Goodfellow |
About Hemogenyx Pharmaceuticals plc
Hemogenyx Pharmaceuticals is a publicly traded company (LSE:HEMO) headquartered in London, with its US operating subsidiaries, Hemogenyx Pharmaceuticals LLC and Immugenyx LLC, located in New York City .
The Company is a clinical stage biopharmaceutical group developing new medicines and treatments to treat blood and autoimmune diseases. Hemogenyx Pharmaceuticals is developing several distinct and complementary product candidates, as well as platform technologies that it uses as engines for novel product development.
This information is provided by RNS, the news service of the London Stock Exchange. RNS is approved by the Financial Conduct Authority to act as a Primary Information Provider in the United Kingdom. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com.
SOURCE: Hemogenyx Pharmaceuticals PLC
View the original press release on ACCESS Newswire
Th.Berger--AMWN