-
 Muchova battles past Svitolina to book Stuttgart final berth
Muchova battles past Svitolina to book Stuttgart final berth
-
Allegri rules out taking Italy job, wants to stay at AC Milan

-
 Miller bludgeons Delhi to IPL win over Bengaluru
Miller bludgeons Delhi to IPL win over Bengaluru
-
Pope says he regrets his remarks interpreted as a debate with Trump

-
 Brentford blow chance for top six in Fulham stalemate
Brentford blow chance for top six in Fulham stalemate
-
Trade ships hit in Hormuz as Iran reopening falters

-
 France blames Hezbollah for French peacekeeper's death in Lebanon
France blames Hezbollah for French peacekeeper's death in Lebanon
-
Venezuela's Machado doesn't regret gifting Nobel Peace Prize to Trump

-
 No date set for next round of Iran-US talks: Iran deputy FM
No date set for next round of Iran-US talks: Iran deputy FM
-
Iran closes Hormuz Strait again over US blockade, ships reverse course

-
 'We've already beaten other favorites', Lyon's Endrick warns PSG
'We've already beaten other favorites', Lyon's Endrick warns PSG
-
Turkey says Israel using security as a pretext to acquire 'more land'

-
 Iran closes Hormuz Strait again over US blockade with ships mid-transit
Iran closes Hormuz Strait again over US blockade with ships mid-transit
-
French film star Nathalie Baye dead at 77: family to AFP

-
 China sex toy makers cautiously embrace AI wave
China sex toy makers cautiously embrace AI wave
-
Paramount's CinemaCon charm offensive gets lukewarm reception

-
 Game over: Players press EU to ban 'destroying' video titles
Game over: Players press EU to ban 'destroying' video titles
-
Churches to the rescue of Cuba's legions of poor

-
 In Trump era, fearful left-leaning Americans turn to guns
In Trump era, fearful left-leaning Americans turn to guns
-
Pope brings Africa tour to Angola as Trump feud drags on

-
 New to The Street to Broadcast Tonight on Bloomberg at 6:30 PM EST - Show #744 Featuring Virtuix Holdings (NASDAQ:VTIX), Neonc Technologies Holdings (NASDAQ:NTHI), Medicus Pharma (NASDAQ:MDCX), YY Group Holding (NASDAQ:YYGH), and Vivos Therapeutics (NASDAQ:VVOS)
New to The Street to Broadcast Tonight on Bloomberg at 6:30 PM EST - Show #744 Featuring Virtuix Holdings (NASDAQ:VTIX), Neonc Technologies Holdings (NASDAQ:NTHI), Medicus Pharma (NASDAQ:MDCX), YY Group Holding (NASDAQ:YYGH), and Vivos Therapeutics (NASDAQ:VVOS)
-
MasterChef Junior Finalist and Fort Lauderdale's Own Remy Powell Debuts #1 New Release Cookbook with Live Event - April 28

-
 Fitzpatrick charges to one-shot lead at RBC Heritage
Fitzpatrick charges to one-shot lead at RBC Heritage
-
Andreeva sinks Swiatek to meet top seed Rybakina in Stuttgart semis

-
 Carrick won't rule out Rashford return to Man Utd
Carrick won't rule out Rashford return to Man Utd
-
Lampard restores reputation by leading Coventry to Premier League

-
 'Gouged': World Cup fans to pay 'insane' $150 for NY stadium train ticket
'Gouged': World Cup fans to pay 'insane' $150 for NY stadium train ticket
-
Lens leave it late to edge Toulouse and keep pressure on PSG

-
 Inter swat aside Cagliari to continue Serie A title procession
Inter swat aside Cagliari to continue Serie A title procession
-
Thunder stay in the moment as NBA title repeat beckons

-
 US Catholics unsettled by Trump's feud with pope
US Catholics unsettled by Trump's feud with pope
-
US Supreme Court sides with Chevron in environmental case

-
 World Cup fans to pay $150 for NY stadium train ticket: official
World Cup fans to pay $150 for NY stadium train ticket: official
-
Gujarat's Gill consigns Kolkata to fifth defeat in IPL

-
 Top takeaways from CinemaCon: the year's hottest movies
Top takeaways from CinemaCon: the year's hottest movies
-
Lebanon president says working on 'permanent agreements' after Israel truce

-
 Top-seeded Pistons embrace underdog tag
Top-seeded Pistons embrace underdog tag
-
Andreeva sinks Swiatek to reach Stuttgart semis

-
 Genital mutilation: the silent suffering of Colombia's Indigenous girls
Genital mutilation: the silent suffering of Colombia's Indigenous girls
-
UEFA probe after photographers injured at Bayern-Real game

-
 Trump tells AFP 'no sticking points' for deal with Iran
Trump tells AFP 'no sticking points' for deal with Iran
-
Trump tells AFP Iran deal close, 'no sticking points' left

-
 Shippers eye Iran Hormuz reopening with wariness
Shippers eye Iran Hormuz reopening with wariness
-
France, UK to lead 'defensive' force for Hormuz

-
 Fils takes out Musetti to reach Barcelona Open semis
Fils takes out Musetti to reach Barcelona Open semis
-
Griezmann soaking up last Atletico moments before 'joy' of Copa final

-
 Polish stadium cancels Kanye West concert
Polish stadium cancels Kanye West concert
-
Lille's Bentaleb out after 'minor surgery' for infection

-
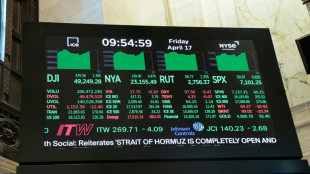 Oil plunges, stocks jump as Iran declares Hormuz open
Oil plunges, stocks jump as Iran declares Hormuz open
-
Trump signals Iran deal near, hails 'brilliant day for world'


US medical agency will scale back testing on monkeys
The United States will scale back certain drug-safety testing requirements on monkeys, federal regulators said Tuesday, marking the latest step by President Donald Trump's administration to limit the use of animals in research.
Under new draft guidance from the Food and Drug Administration, computer models, lab-grown mini-organs, and human studies will replace the use of non-human primates in testing monoclonal antibodies -- lab-engineered proteins used to treat cancers, autoimmune conditions and other diseases.
"We are delivering on our roadmap commitment to eliminate animal testing requirements in drug evaluation and our promise to accelerate cures and meaningful treatments for Americans," FDA Commissioner Marty Makary said in a statement.
The statement added that typical nonclinical programs involving monoclonal antibodies could include more than 100 macaque monkeys -- apes are no longer used in any invasive research in the US -- yet often do not yield human-approved treatments.
The move was welcomed by animal-advocacy groups.
Zaher Nahle, a former animal researcher who is now the senior scientific advisor for nonprofit Center for a Humane Economy, told AFP the move was an "important step."
"These primates are not reliable in terms of predicting the toxicity, so you can get at least equal or better results in terms of your accuracy in predicting toxicology using other approaches," he added.
What's more, he noted, studies show that more than 90 percent of drugs deemed safe and effective in animals fail to win approval for human use.
The FDA's announcement follows a report in the journal Science last month that the Centers for Disease Control and Prevention would close its primate labs.
It "moves us one step closer to wiping out the federal government's wasteful monkey business," Justin Goodman of White Coat Waste Project told AFP.
But the National Institutes of Health -- the country's primary biomedical research agency -- remains a notable "outlier," he added. According to public data, 7,700 primates are confined in federal government labs and breeding facilities, of which 6,700 are at NIH.
Advocates of animal research have pushed back.
"Poorly planned transitions -- including premature study termination -- can place animals at serious risk and may undermine years of scientific investment intended to strengthen public health preparedness and advance lifesaving medical progress," advocacy group Americans for Medical Progress said after the CDC news was reported.
Proponents of animal testing say the research has been indispensable for major medical advances, including vaccines for diphtheria, yellow fever, measles and Covid-19.
Critics counter that decades-old laws have created regulatory lock-in, that publication incentives reward animal studies in top journals, and that a lucrative "animal-industrial complex" has helped entrench the status quo.
D.Kaufman--AMWN